Patents
Literature
Hiro is an intelligent assistant for R&D personnel, combined with Patent DNA, to facilitate innovative research.
1 results about "Reaction site" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
An injection site reaction is inflammation in or damage to the tissue surrounding where a drug was injected. There are two types of injection site reactions: a local allergic reaction called a flare reaction, and a more severe reaction characterized by damage to the tissue due to extravasation.
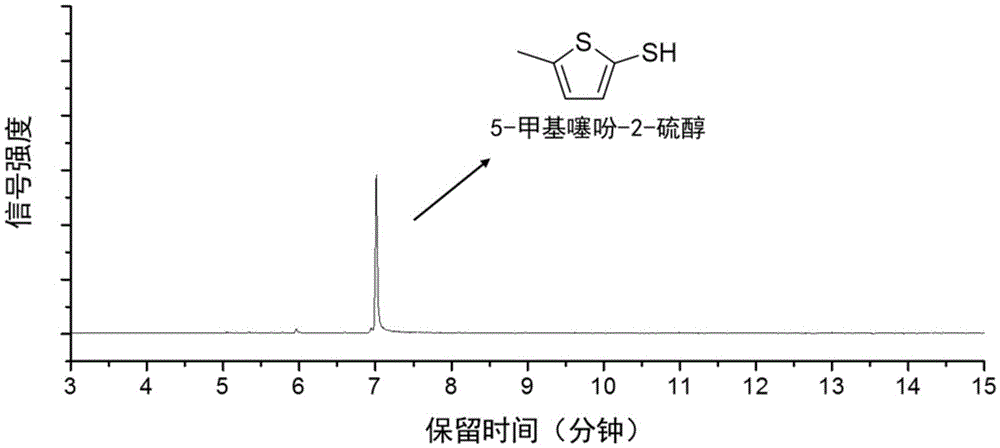
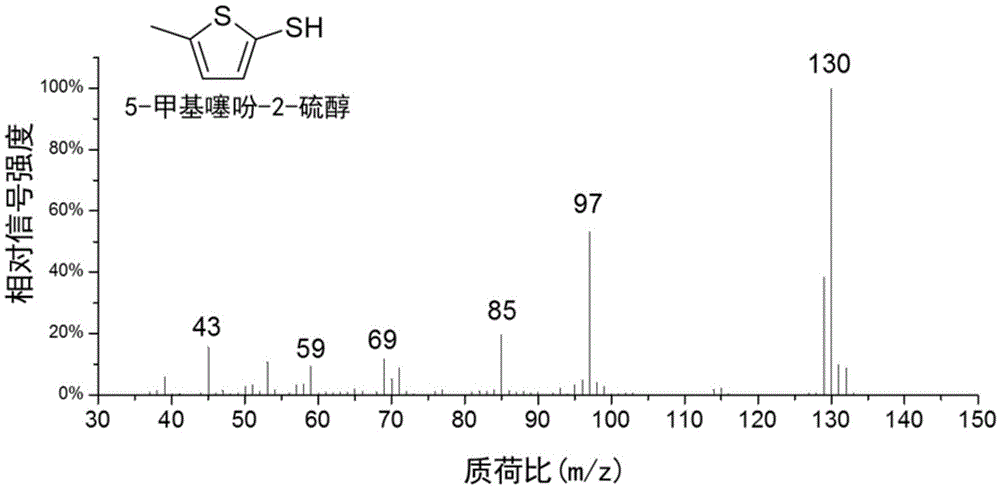
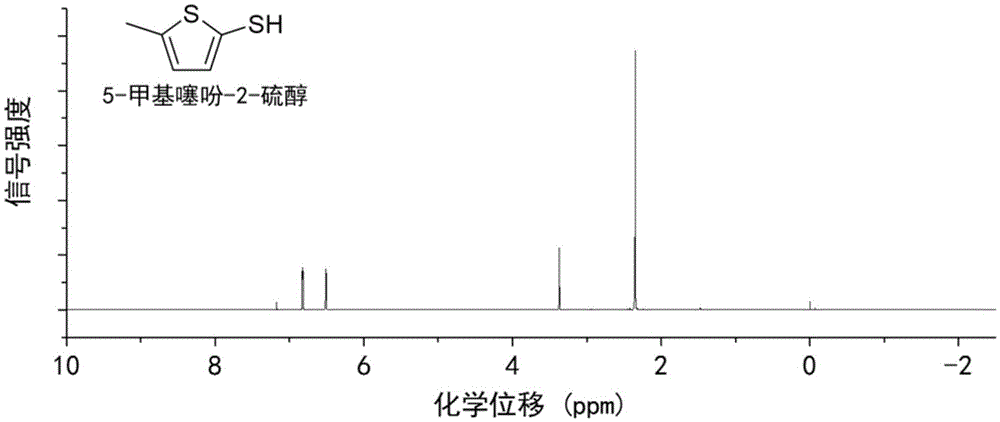
Method for preparing 5-methylthienyl-2-thiol from levulinic acid
Owner:XIAMEN UNIV
Popular searches
Who we serve
- R&D Engineer
- R&D Manager
- IP Professional
Why Eureka
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Social media
Try Eureka
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap