Method for assisting diagnosis of risk of progression to nephropathy and use of reagent kit
A technology for kidney disease and risk, which is applied in the field of auxiliary diagnosis of kidney disease development risk, and can solve problems such as reduction of medical expenses
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
Embodiment approach
[0029] In the method of diagnosing the risk of developing nephropathy (hereinafter also referred to as "diagnosing method") of the present embodiment, first, the urinary apolipoprotein A1 (ApoA1) concentration of a diabetic patient with prenephropathy is measured.
[0030] In the diagnostic method of this embodiment, a diabetic patient with pre-renal disease (hereinafter also referred to as a "test subject") is targeted for diagnosis. Pre-nephropathy is one of the stages of diabetic nephropathy, also known as stage 1. Currently, the stage classification of diabetic nephropathy refers to the severity classification of chronic kidney disease (CKD), as shown in Table 1 below.
[0031] 【Table 1】
[0032]
[0033] In this specification, pre-renal disease and stage 1 mean that the albuminuria classification in the CKD severity classification described in KDIGO (Kidney Disease: Improving Global Outcomes) 2012 Clinical Practice Guideline for the Evaluation and Management of Chronic K
Embodiment 1
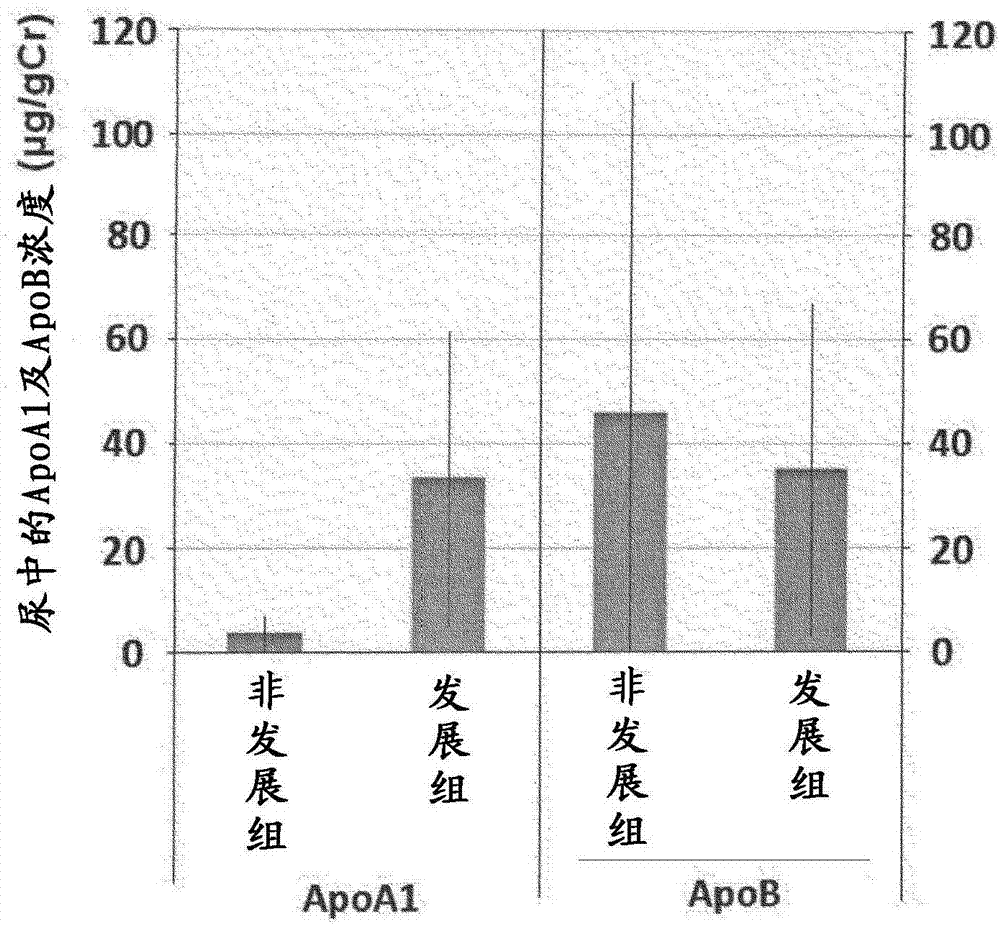
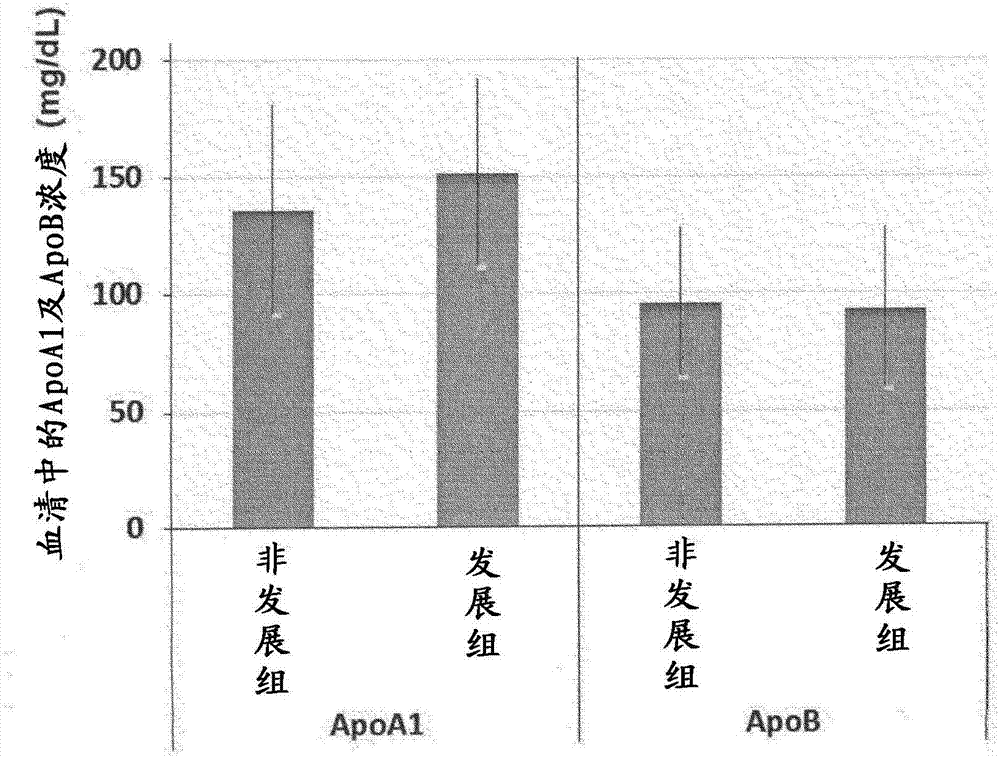
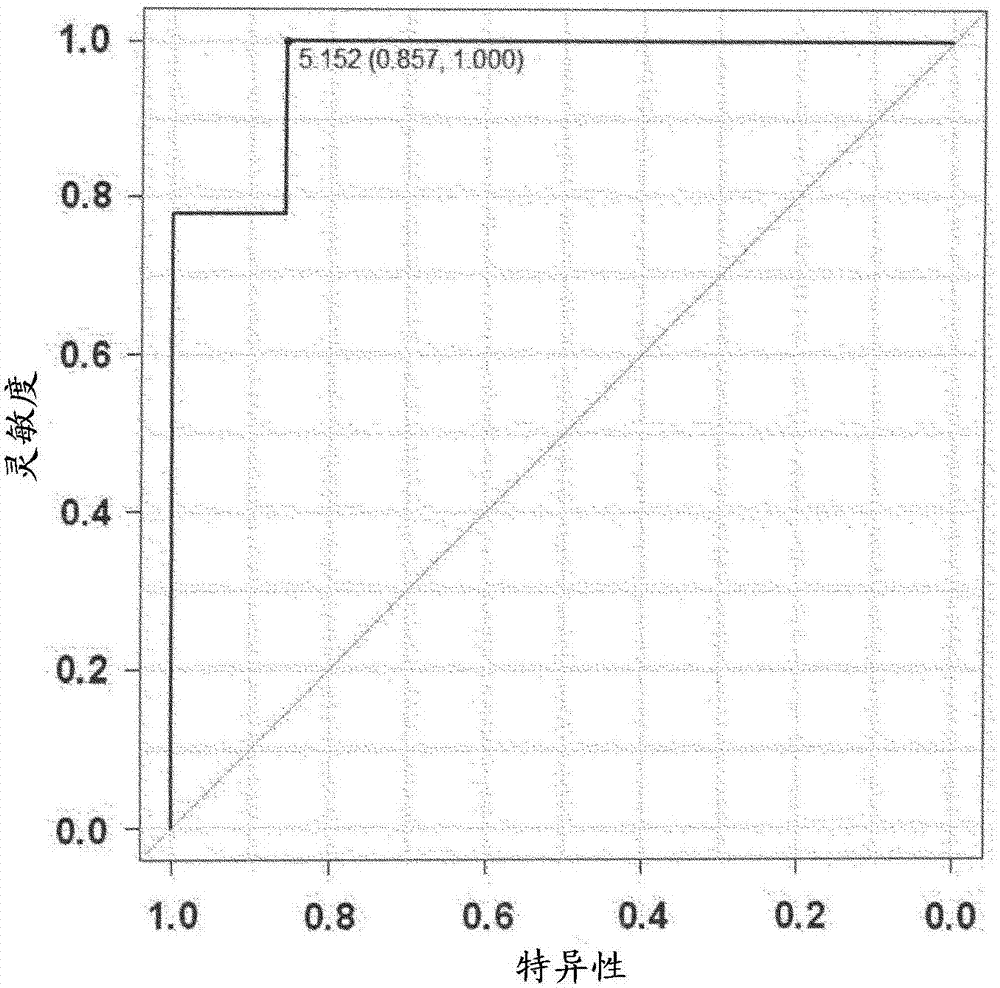
[0066] Blood and urine were collected from subjects who were diabetic patients with pre-nephropathy (stage 1), and after 6 months, these subjects were divided into a group that developed nephropathy and a group that did not develop nephropathy. The concentrations of ApoA1 and ApoB in the blood and urine of the test subjects in each group were compared. Furthermore, ApoB is known as a biomarker of kidney disease.
[0067] (1) Collection of samples from subjects
[0068] From urinary albumin / Cr ratio below 30mg / gCr, and GFR differentiated at 30mL / min / 1.73m 2 Blood and urine were collected as samples from the above diabetic patients (n=14).
[0069] (2) Determination of biomarkers
[0070] (2.1) Determination of the concentration of ApoA1, ApoB and creatinine in serum
[0071] Using ApoA-IAUT·N "first" and ApoBAUT·N "first" (both obtained from Sekisui Medical Co., Ltd.), ApoA1 and ApoA1 in serum were measured from the blood of each subject obtained in (1) above. Concent
Embodiment 2
[0084] For subjects who are diabetic patients in the early stage of nephropathy (stage 2) and overt stage of nephropathy (stage 3), it was also investigated whether the risk of developing nephropathy can be predicted by the concentration of ApoA1 in urine.
[0085] (1) Collection of samples from subjects
[0086] From urinary albumin / Cr ratio below 30mg / gCr to above 300mg / gCr, and GFR differentiated at 30mL / min / 1.73m 2 Diabetic patients above (stage 2, n=6) with urinary albumin / Cr ratio above 300mg / gCr and GFR at 30mL / min / 1.73m 2 Blood and urine were collected as samples from the above diabetic patients (third stage, n=8).
[0087] (2) Determination of biomarkers
[0088] Urine ApoA1 concentration, serum and urine creatinine concentrations were measured in the same manner as in Example 1. In addition, the urinary ApoA1 concentration per 1 g of creatinine was calculated from the urinary ApoA1 concentration and the urinary creatinine concentration.
[0089] (3
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap