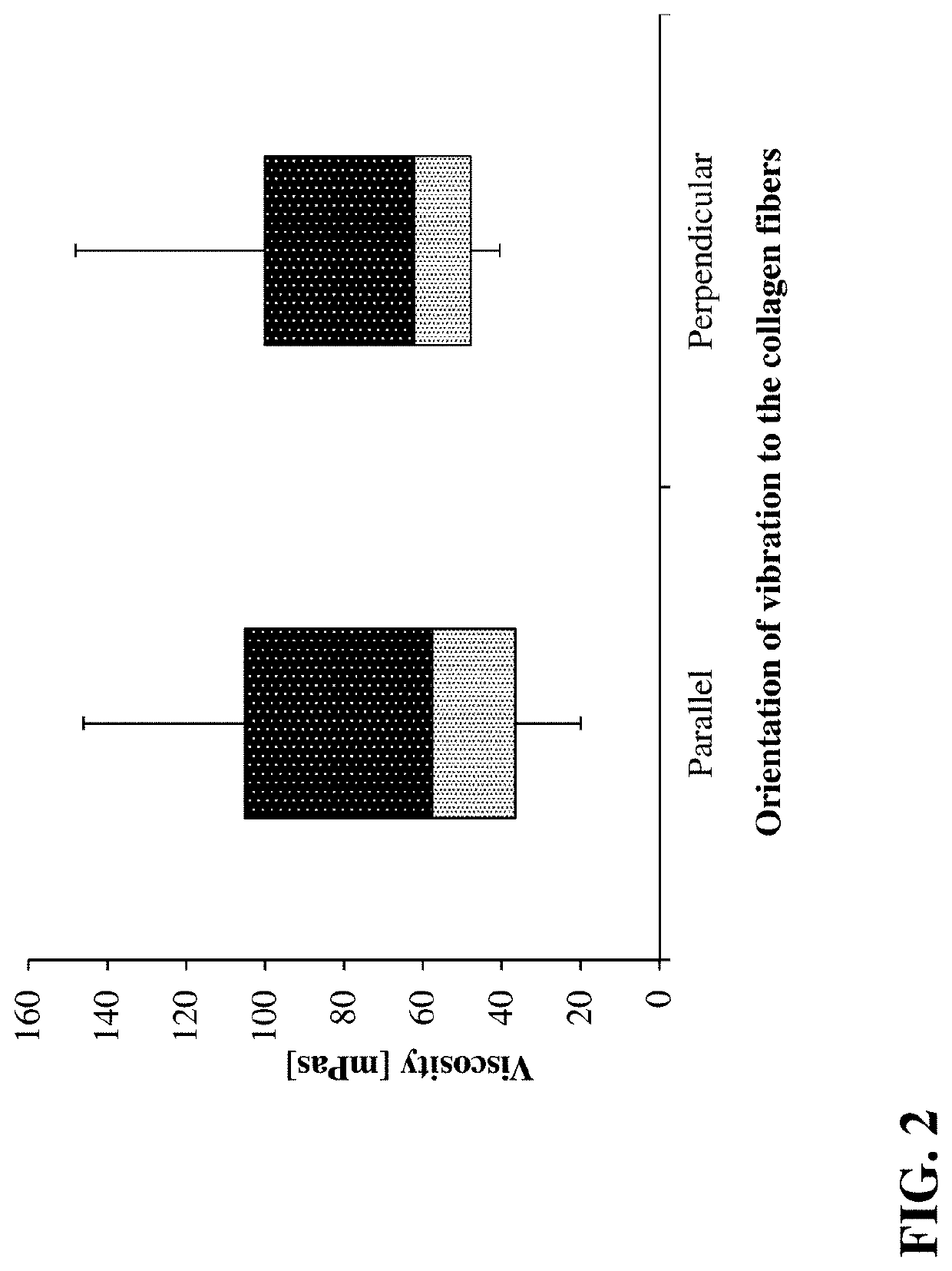
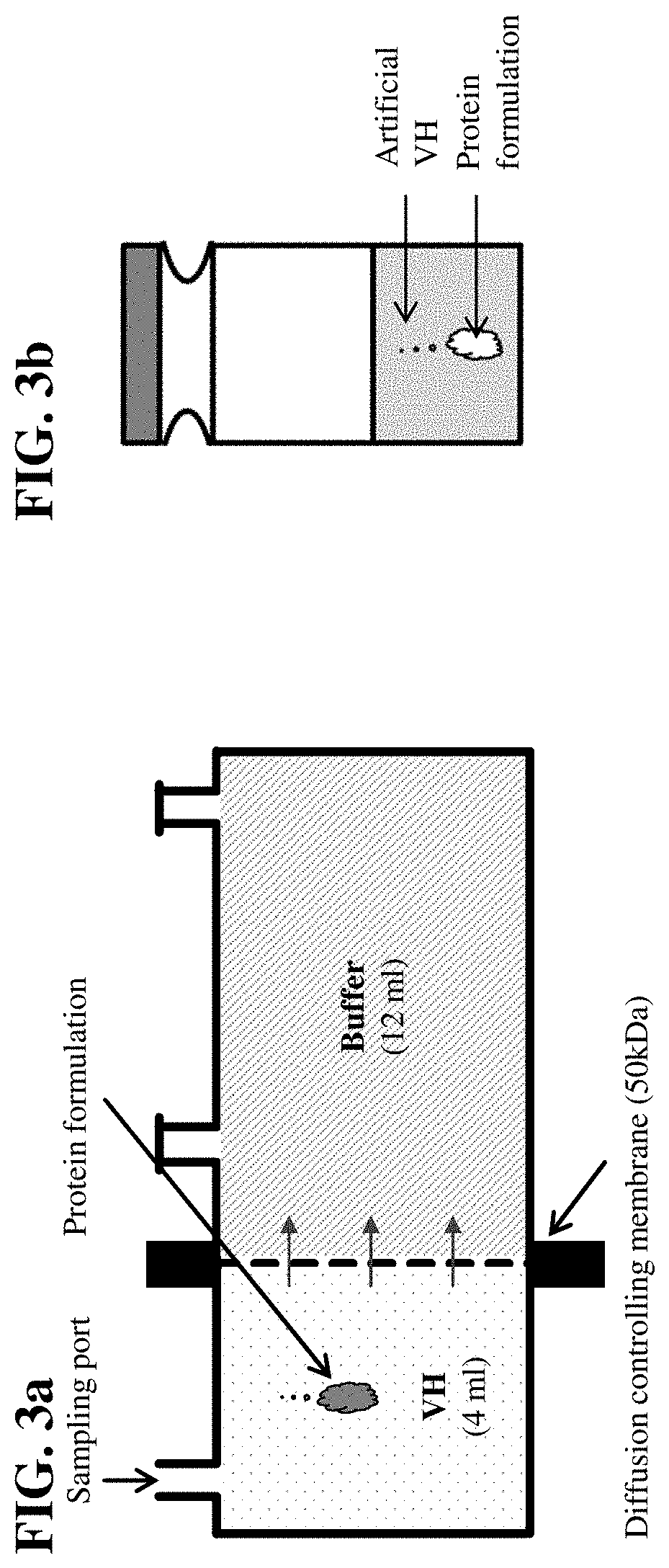
Artificial Vitreous Humor for the Investigation of Drugs and Drug Formulations
a technology of vitreous humor and drug formulation, which is applied in the direction of pharmaceutical delivery mechanism, macromolecular non-active ingredients, inorganic non-active ingredients, etc., can solve the problems that the test may not represent the actual situation in the eye of a living subject, and the in vitro test to study for example the stability of a drug formulation in vh has proved little useful when simulating real in vivo conditions
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
[0046]Development of a Simulated Buffer Medium (SBM)
[0047]Natural VH is a liquid tissue mainly composed of water with small amounts of organic and inorganic components including matrix forming polymers. The composition of VH is reported in literature. In order to prepare artificial VH (aVH), firstly, it was important to identify a base buffer system which can support the physicochemical stability of aVH. Secondly, it was important to prepare a simulated buffer medium (SBM) containing all the small molecular weight components of the natural VH in a defined concentration as reported in literature. At last, SBM was prepared in the presence of matrix forming polymers i.e., hyaluronic acid and collagen, inherent components of natural VH. Stability of the aVH was then evaluated by storing it at 37° C. for 3 months.
[0048]In order to identify the right buffer system, two biologically relevant buffer systems supporting physiological pH such as Kerbs Ringer bicarbonate buffer and phosphate buffe
example 2
[0053]Impact of Higher Buffer Strength, Sodium Lactate, Hypoxanthine and Ascorbic Acid on the Stability of SBMs
[0054]In order to find stable composition, SBM with or without sodium lactate, hypoxanthine and ascorbic acid, and SBM with higher buffer strength were investigated. The SBMs were prepared as reported in Table 5.
TABLE 5Impact of buffer strength and presence / absence of sodium lactate, hypoxanthine, andascorbic acid on the storage stability of simulated buffer medium (SBM) at 37° C.Ascorbic GlucoseUreaSodium PBSHypoxanthineCreatinineXanthineacid (350 lactateGlutathionepHpH#(pH 7.4)(309 μM)(64.6 μM)(580 μM)μg / mL)(2.2 mM)(7.6 mM)(10.5 mM)(200 μM)Initial7 days10 0.01M++++++++PhysiologicalAcidic180.015M++++++++PhysiologicalAcidic19 0.02M++++++++PhysiologicalAcidic20 0.04M++++++++PhysiologicalAcidic210.015M+++++++PhysiologicalAcidic22 0.02M+++++++PhysiologicalAcidic230.015M+++++++PhysiologicalAcidic24 0.02M+++++++PhysiologicalAcidic25 0.01M++++++PhysiologicalAcidic260.015M++++++P
example 3
[0057]Long Term Stability of SBM
[0058]Once the composition 28 of SBMs was found stable for 7 days without change in pH or clarity, it was further characterized for long-term storage stability at 37° C. Briefly, composition 28 was prepared and aseptically filtered under LAF. The sterile SBM was then transferred aseptically in 6 mL sterile glass vials. The glass vials were sealed and incubated at 37° C. for 3 months. The samples were collected at various time points such as, initial, week-1, week-2, week-5, week-8 and week-12, and analyzed for pH, clarity, density, viscosity and osmolality. For pH estimation, samples (100 μL) were aseptically transferred in eppendorf tubes and analyzed by calibrated pH meter (827 pH Lab, Metrohm, Switzerland). Clarity of the SBMs was observed visually and the samples were classified as clear or turbid. Density of the SBM was estimated at 20° C. using DMA 38 (Anton Paar) densiometer. Osmolality of the samples were estimated with a calibrated osmometer (Os
PUM
| Property | Measurement | Unit |
|---|---|---|
| Temperature | aaaaa | aaaaa |
| Time | aaaaa | aaaaa |
| Mass | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap