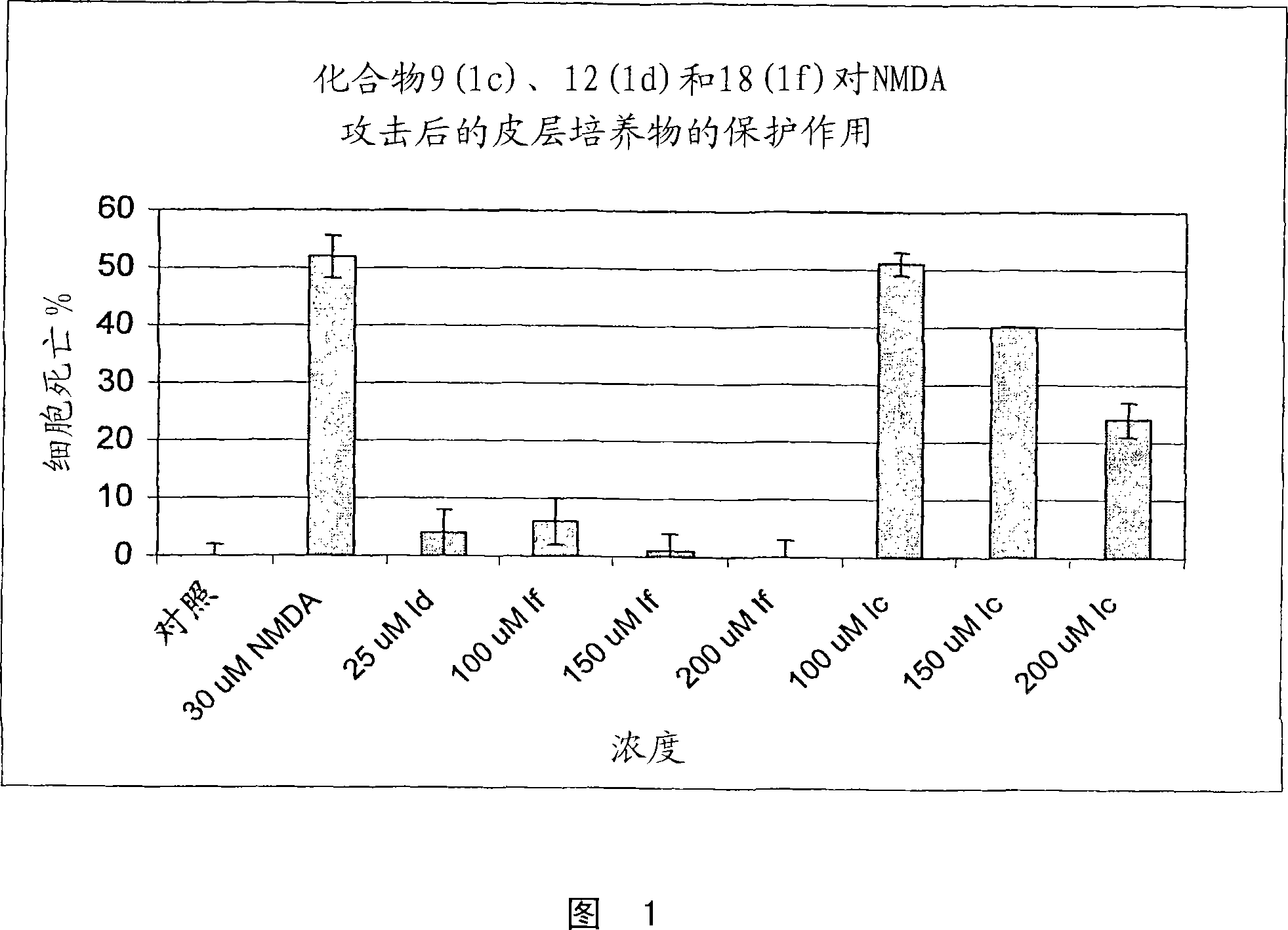
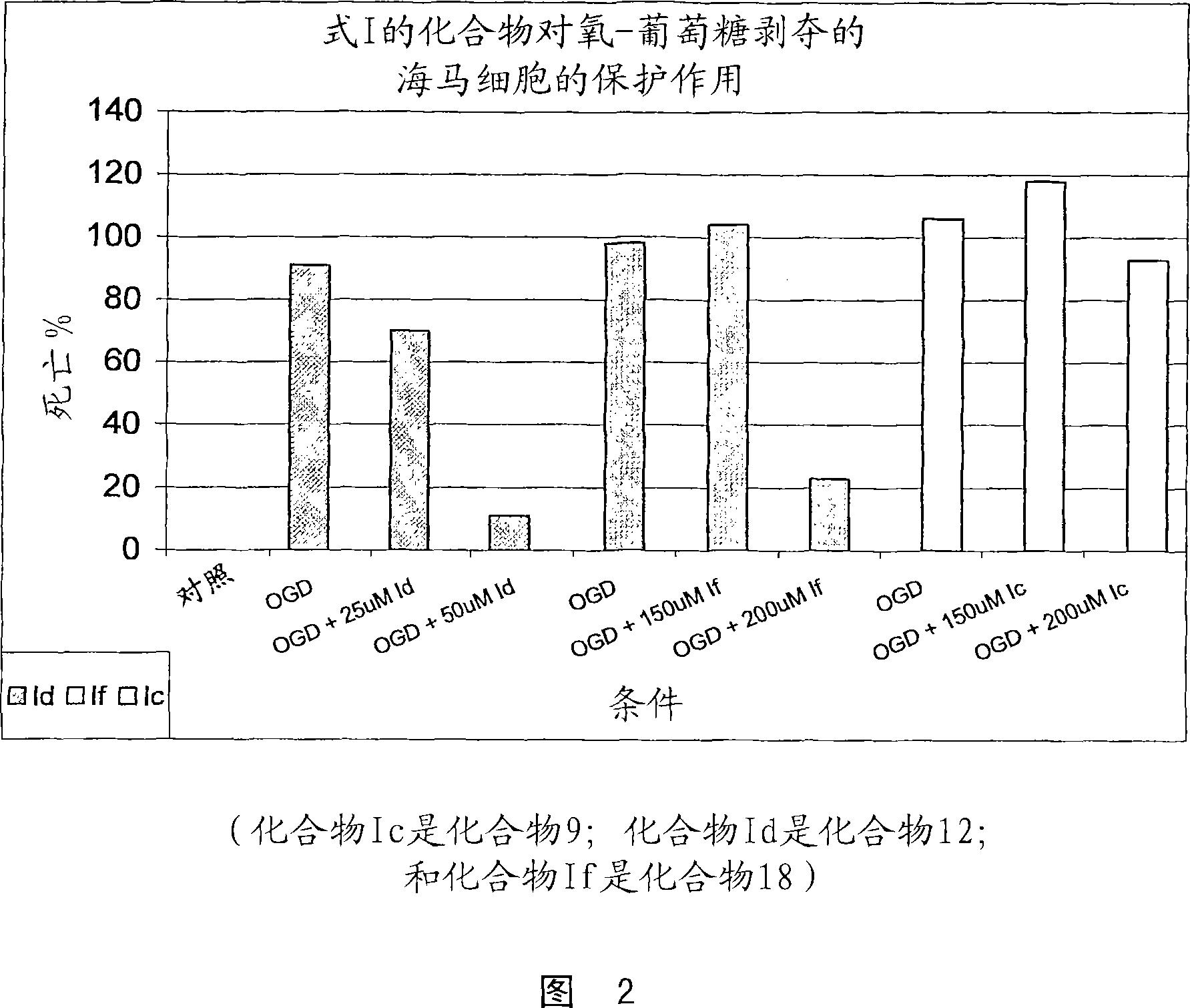
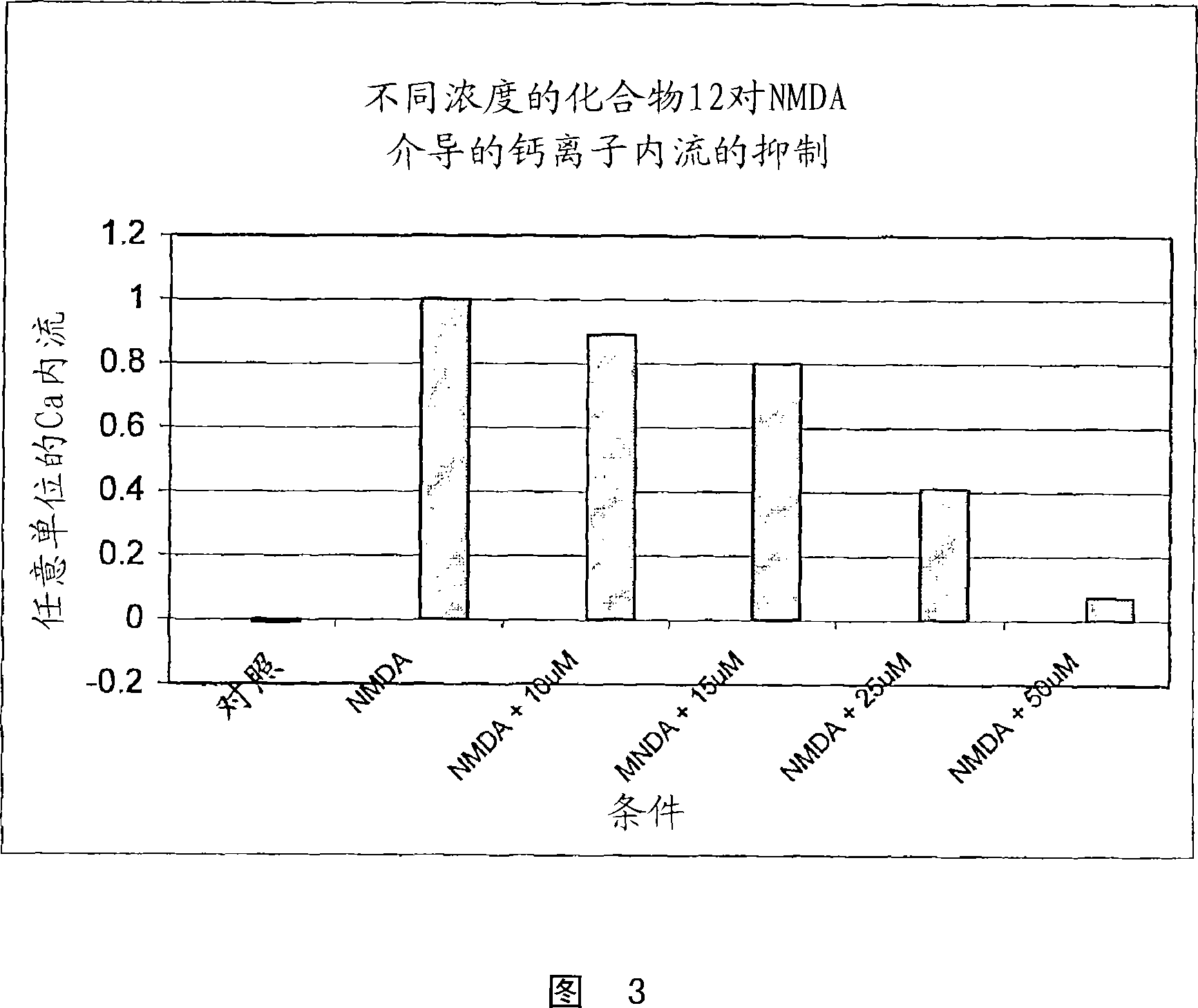
Substituted indole compounds having NOS inhibitory activity
Technology of a compound, thioalkoxy, in the field of substituted indole compounds with NOS inhibitory activity
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0297] Embodiment 1: the preparation of compound 4
[0298]
[0299] (a) Preparation of compound 2: 1H-indol-5-ylamine (compound 1, 100 mg, 0.757 mmol) was dissolved in anhydrous tetrahydrofuran (4.5 mL) in an argon-purged small flask. Benzoyl isothiocyanate (123 mg, 0.757 mmol) was added dropwise and the reaction was stirred at room temperature under argon for 60 hours. 3-(Diethylenetriamino)propyl-functionalized silica gel (0.5 g) was added, the mixture was stirred for an additional 30 minutes, and the mixture was filtered using 3:7 ethyl acetate / hexane as eluent. Purification by silica gel column chromatography (30% ethyl acetate / hexane) gave the product (compound 2, 90 mg, 40.3% yield); 1 H NMR (CDCl 3 )δ: 6.59(s, 1H), 7.25-7.26(m, 2H), 7.51(s, 1H), 7.54-7.66(m, 3H), 7.93(m, 3H), 8.32(br s, 1H), 9.15(s, 1H), 12.50(s, 1H).
[0300] (b) Preparation of compound 3: 1-benzoyl-3-(1H-indol-5-yl)-thiourea (compound 2, 90 mg, 0.305 mmol) was dissolved in a small flask pur
Embodiment 2
[0302] Embodiment 2: the preparation of compound 5
[0303]
[0304] (a) Preparation of compound 5: 1H-indol-5-ylamine (compound 1, 59 mg, 0.45 mmol) and thiophene-2-thiocarbimidothioic acid phenyl ester hydrobromide (142.7 mg, 0.47 mmol) was dissolved in absolute ethanol (2.0 mL) in an argon-purged dry flask. The reaction was stirred at ambient temperature under argon for 17 hours. The solution was diluted with diethyl ether (20 mL) to form a tan precipitate which was collected, washed with diethyl ether and suction dried to give compound 5 as a tan solid (121.4 mg, HBr salt, 84% yield); 1 H NMR (DMSO-d6) δ: 10.9 (s, 1H, NH), 7.74 (d, 1H, J = 3.4), 7.63 (d, 1H, J = 4.88), 7.35 (d, 1H, J = 8.3) , 7 29(s, 1H), 7.12(t, 1H, J=4.88), 7.03(s, 1H), 6.69(d, 1H, J=8.3), 6.35(br s, 2H), 6.35(s, 1H).
Embodiment 3
[0305] Embodiment 3: the preparation of compound 9
[0306]
[0307] (a) Preparation of compound 7: 6-nitroindole (compound 6, 95mg, 0.59mmol) and 1-(2-chloroethyl) pyrrolidine hydrochloride (100mg, 0.59mmol) were dissolved in DMF (3ml) in the swept flask. The reaction was placed in an oil bath preheated to 50 °C and stirred in the presence of potassium carbonate (244 mg, 1.77 mmol) under argon for 24 hours. After cooling, the reaction vessel was placed in an ice bath, and the reaction was diluted with ice water (10 mL) and ethyl acetate. The reaction was transferred to a separatory funnel and the organic layer was collected. The organic layer was washed twice with brine, and the combined aqueous washes were re-extracted with ethyl acetate. The combined organic extracts were dried over sodium sulfate, filtered and concentrated to give a yellow oil. The product was taken up in methanol (2 mL) and acidified with 2N HCl (15 mL), then filtered to remove any insoluble materia
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap