Porcine reproductive and respiratory syndrome virus antibody detection kit and application thereof
A respiratory syndrome and antibody detection technology, applied in biological testing, measuring devices, immunoassays, etc., to achieve the effect of short time, high sensitivity, and simple operation
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0019] The anti-fluorescein FITC antibody-labeled magnetic microparticle solution, wherein the anti-fluorescein FITC antibody is coupled with the anti-FITC antibody by a two-step glutaraldehyde method, and contains 0.5% BSA, 1% casein, The blocking was completed with phosphate buffer at pH 7.2 (0.02 mol / L).
[0020] The preparation of the fluorescein FITC-labeled porcine reproductive and respiratory syndrome virus antigen solution includes the following steps:
[0021] S1: Mix FITC and porcine reproductive and respiratory syndrome virus at 1:1, and react at 37°C in the dark for 12 hours to form a labeling solution.
[0022] S2: dialyze the labeled solution obtained in S1 with 0.01MPBS at 4°C for 24h, add an equal volume of glycerol, and store at -20°C.
[0023] The present invention also provides an application of the porcine reproductive and respiratory syndrome virus antibody detection kit, characterized in that it includes the following steps:
[0024] S1: Put the sample r
Embodiment 2
[0032] specific experiment
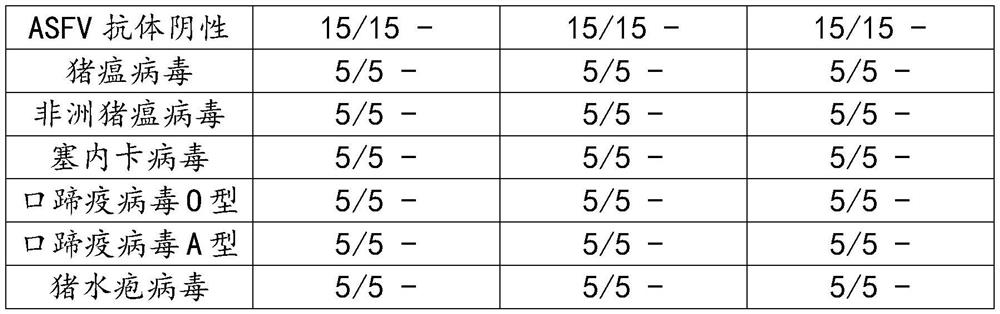
[0033] Three batches of prepared ASFV antibody magnetic particle chemiluminescence kits were used to detect ASFV antibody positive serum (20); ASFV antibody negative serum (15); swine fever virus positive serum 5; African swine fever virus positive serum 5; 5 Seneca virus positive sera; 5 foot-and-mouth disease virus O-positive sera; 5 foot-and-mouth disease virus A-positive sera; porcine vesicular virus positive sera (5); a total of 65 sera were tested, according to the measured luminescence value Calculate S / N to determine negative and positive.
[0034] Table 1 Specificity test of porcine reproductive and respiratory syndrome virus antibody detection kit
[0035]
[0036]
[0037] The results showed that the "Swine Reproductive and Respiratory Syndrome Virus Antibody Detection Kit" did not cross-react with the positive serum of swine fever, porcine reproductive and respiratory syndrome, Seneca, foot-and-mouth disease type O, foot-and-mouth
Embodiment 3
[0039] Sensitivity test
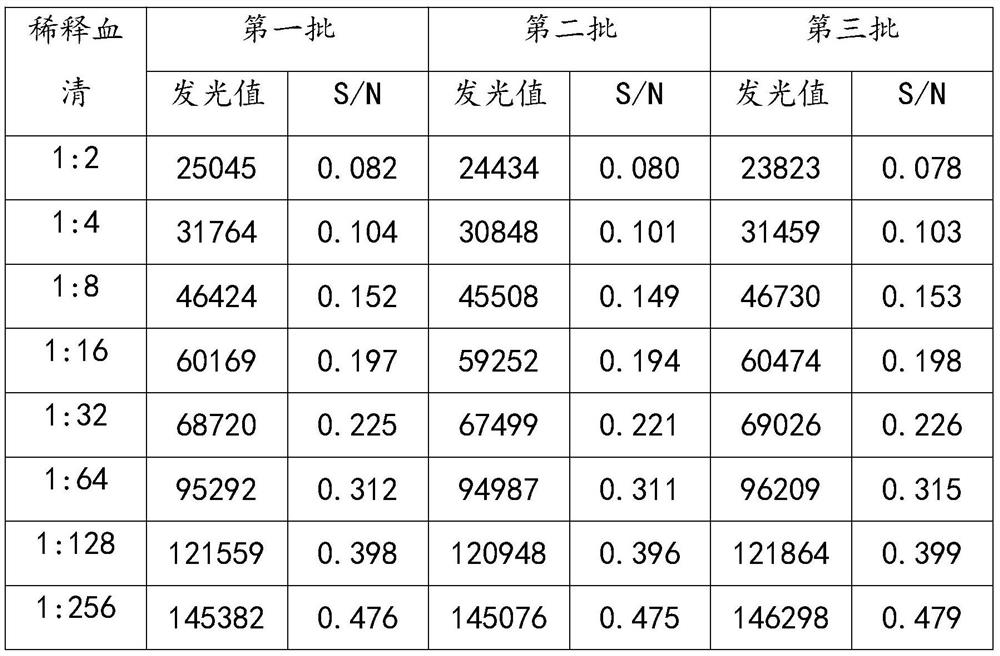
[0040] Dilute the positive standard serum with the sample diluent at 1:2, 1:4, 1:8, 1:16, 1:32, 1:64, 1:128, 1:256, 1:512, 1:1024, It was diluted at a ratio of 1:2048 and used as a sensitive quality control serum for detection with three batches of ASFV magnetic particle chemiluminescence kits prepared.
[0041] Table 2 Sensitivity test of porcine reproductive and respiratory syndrome virus antibody detection kit
[0042]
[0043]
[0044] The test results showed that the data of the three repeated experiments were consistent, and the minimum detection amount of porcine reproductive and respiratory syndrome antibody could reach 1:256.
PUM
| Property | Measurement | Unit |
|---|---|---|
| Ph | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap