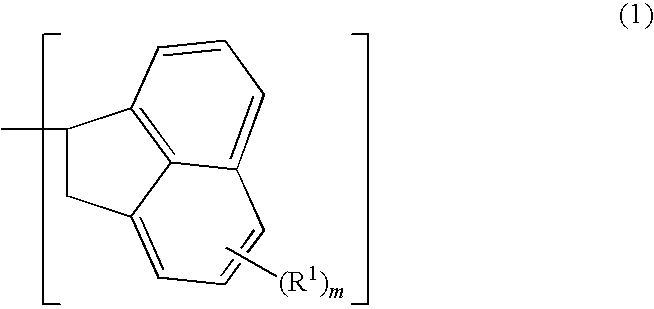
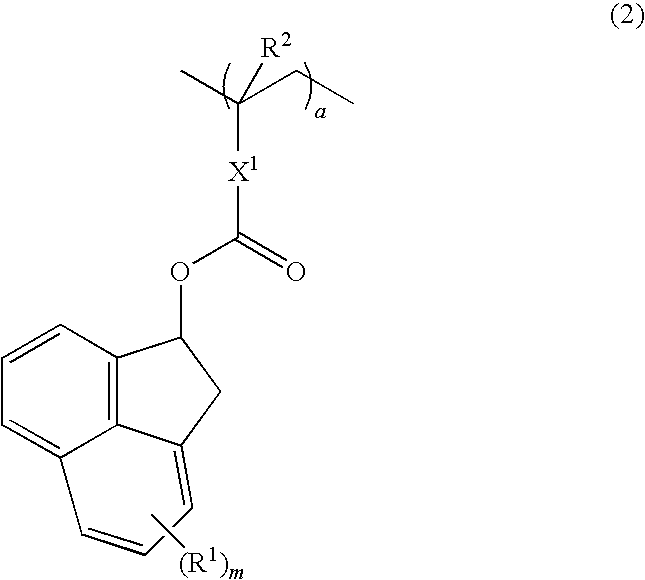
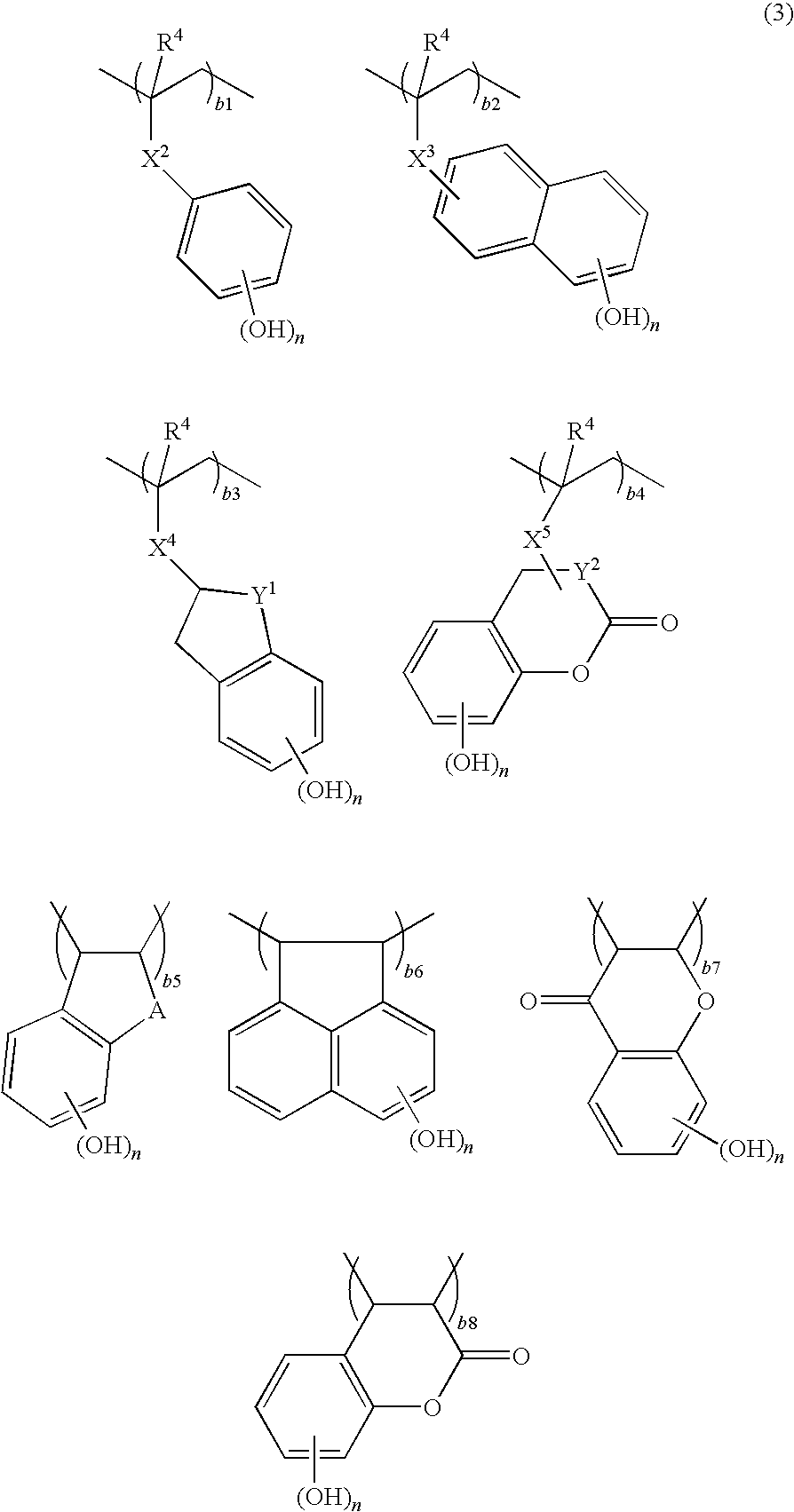
Patents
Literature
Hiro is an intelligent assistant for R&D personnel, combined with Patent DNA, to facilitate innovative research.
2 results about "Acid labile" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
In the context of chemistry, acid labile means that an acidic proton is readily formed. For example, this can occur in molecules with a hydroxyl group with conjugated double bond capable of stabilising the anionic charge from extra electrons. This makes the acidic H+ easier to release, or more labile. Related QuestionsMore Answers Below.
Positive resist composition and patterning process
ActiveUS20100227273A1Increase contrastGood pattern profilePhotosensitive materialsRadiation applicationsPhotochemistryAcid labile
Owner:SHIN ETSU CHEM IND CO LTD
Functionalized carbosilane polymers and photoresist compositions containing the same
Linear or branched functionalized polycarbosilanes having an absorbance less than 3.0 μm−1 at 193 nm and a relatively high refractive index are provided. The functionalized polycarbosilanes contain at least one pendant group that is acid labile or aqueous base soluble. Also disclosed are photoresists formulations containing the functionalized polycarbosilanes that are suitable for use in lithography, e.g., immersion lithography.
Owner:GLOBALFOUNDRIES INC
Popular searches
Who we serve
- R&D Engineer
- R&D Manager
- IP Professional
Why Eureka
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Social media
Try Eureka
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap