Patents
Literature
Hiro is an intelligent assistant for R&D personnel, combined with Patent DNA, to facilitate innovative research.
1 results about "Hydroxamic acid" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
A hydroxamic acid is a class of organic compounds bearing the functional group RC(O)N(OH)R', with R and R' as organic residues and CO as a carbonyl group. They are amides (RC(O)NHR') wherein the NH center has an OH substitution. They are often used as metal chelators.
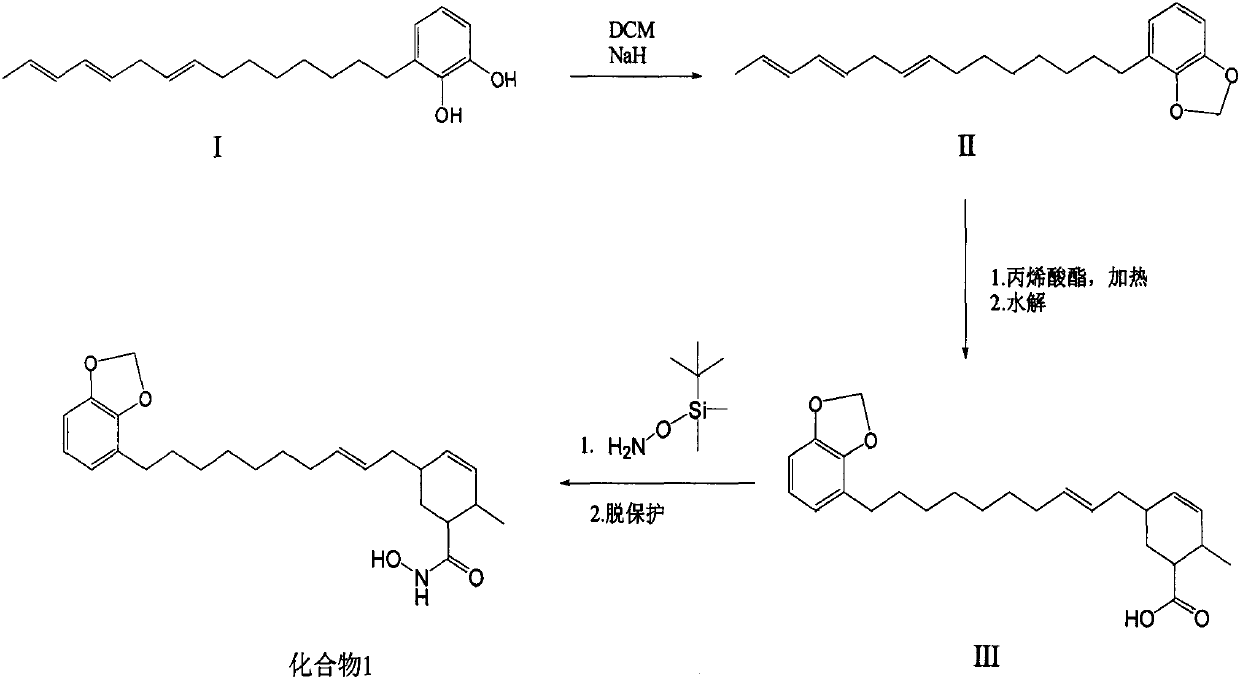
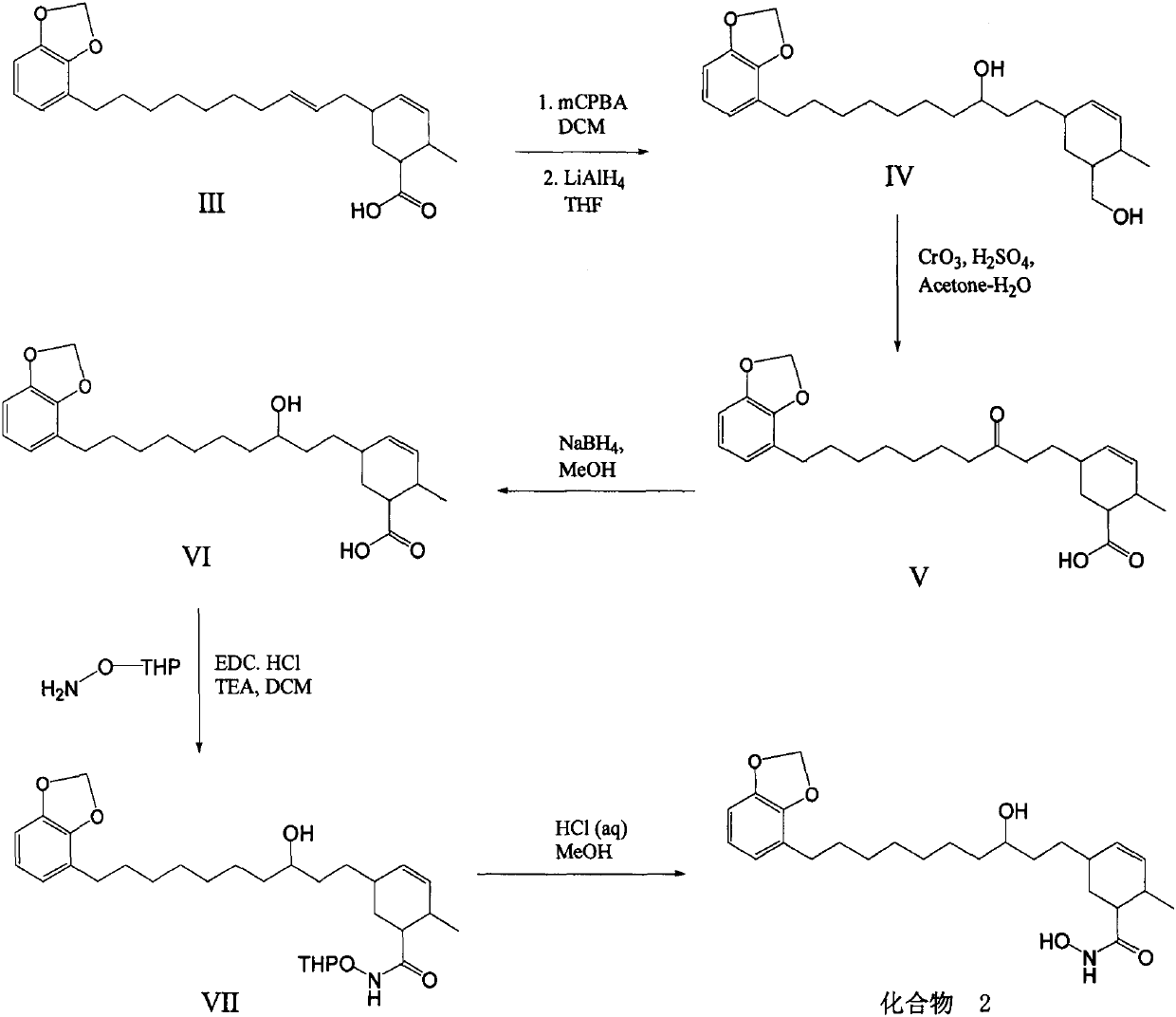
Synthetic method of methylene ether urushiol hydroxamic acid derivatives with (histone deacetylase) HDAC inhibitory activity
ActiveCN110256398AWide range of sourcesOrganic chemistryAntineoplastic agentsSide chainHydroxamic acid
Owner:INST OF CHEM IND OF FOREST PROD CHINESE ACAD OF FORESTRY
Popular searches
Who we serve
- R&D Engineer
- R&D Manager
- IP Professional
Why Eureka
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Social media
Try Eureka
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap