Aspirin sustained-release tablets and preparation method thereof
A technology of aspirin and sustained-release tablets, which is applied in the direction of pharmaceutical formulas, medical preparations with non-active ingredients, medical preparations containing active ingredients, etc., can solve problems such as poor quality stability, large quality differences, stimulation, etc., and achieve uniform quality Good sex, good stability, safe drug effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
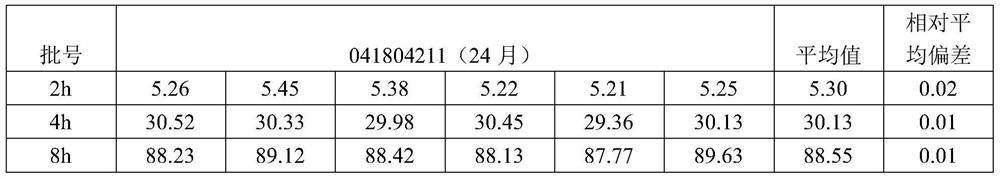
[0017] Take 750 parts of aspirin (the corresponding weight is 75.0g, and the rest of the ingredients follow this ratio), 35 parts of starch, 63 parts of polyacrylic acid resin II, 54 parts of hypromellose, and 8 parts of ethyl cellulose, and grind them through an 80-mesh sieve net, spare.
[0018] 3% hypromellose alcohol solution prescription, including tartaric acid, water, polysorbate 80, hypromellose, 95% ethanol.
[0019] Prepare 3% hypromellose alcohol solution: add 135 parts of tartaric acid to 360 parts of purified water, heat to about 70°C and stir to completely dissolve, then add 36 parts of polysorbate 80, stir to completely dissolve, and obtain solution A; Another 54 parts of hypromellose was added while stirring, and it was evenly dispersed in 1152 parts of ethanol to obtain solution B; the obtained two solutions of A and B were mixed evenly, and placed at 18°C for 8 hours.
[0020] The above tartaric acid, water, polysorbate 80, hypromellose, ethyl cellulose, and
Embodiment 2
[0029] Take 750 parts of aspirin, 30 parts of starch, 50 parts of polyacrylic resin II, 40 parts of hypromellose, and 8 parts of ethyl cellulose, grind them through an 80-mesh sieve, and set aside.
[0030] 3% hypromellose alcohol solution prescription, including tartaric acid, water, polysorbate 80, hypromellose, 95% ethanol.
[0031] Prepare 3% hypromellose alcohol solution: add 100 parts of tartaric acid to 200 parts of purified water, heat to about 70°C and stir to completely dissolve, then add 20 parts of polysorbate 80, stir to completely dissolve, and obtain solution A; Another 54 parts of hypromellose was added while stirring, and it was evenly dispersed in 600 parts of ethanol to obtain solution B; the obtained two solutions of A and B were mixed evenly, and placed at 20°C for 30 hours.
[0032] The above tartaric acid, water, polysorbate 80, hypromellose, ethyl cellulose, and 95% ethanol all should meet the requirements of "The Pharmacopoeia of the People's Republic of
Embodiment 3
[0041] Take 750 parts of aspirin, 50 parts of starch, 70 parts of polyacrylic resin II, 60 parts of hypromellose, and 8 parts of ethyl cellulose, grind them through an 80-mesh sieve, and set aside.
[0042] 3% hypromellose alcohol solution prescription, including tartaric acid, water, polysorbate 80, hypromellose, 95% ethanol.
[0043]Prepare 3% hypromellose alcohol solution: add 150 parts of tartaric acid to 500 parts of purified water, heat to about 70°C and stir to completely dissolve, then add 50 parts of polysorbate 80, stir to completely dissolve, and obtain solution A; Another 60 parts of hypromellose was added while stirring, and it was evenly dispersed in 1500 parts of ethanol to obtain solution B; the obtained two solutions of A and B were mixed evenly, and placed at 26°C for 72 hours.
[0044] The above tartaric acid, water, polysorbate 80, hypromellose, ethyl cellulose, and 95% ethanol all should meet the requirements of "The Pharmacopoeia of the People's Republic of
PUM
| Property | Measurement | Unit |
|---|---|---|
| Viscosity | aaaaa | aaaaa |
| Viscosity | aaaaa | aaaaa |
| Viscosity | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap