Method for preparing pyrazolecarboxylic acid derivative, and intermediate thereof
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
ration Method of 3-(difluoromethyl)-1-methyl-1H-pyrazole-4-carboxylic Acid
[0090]
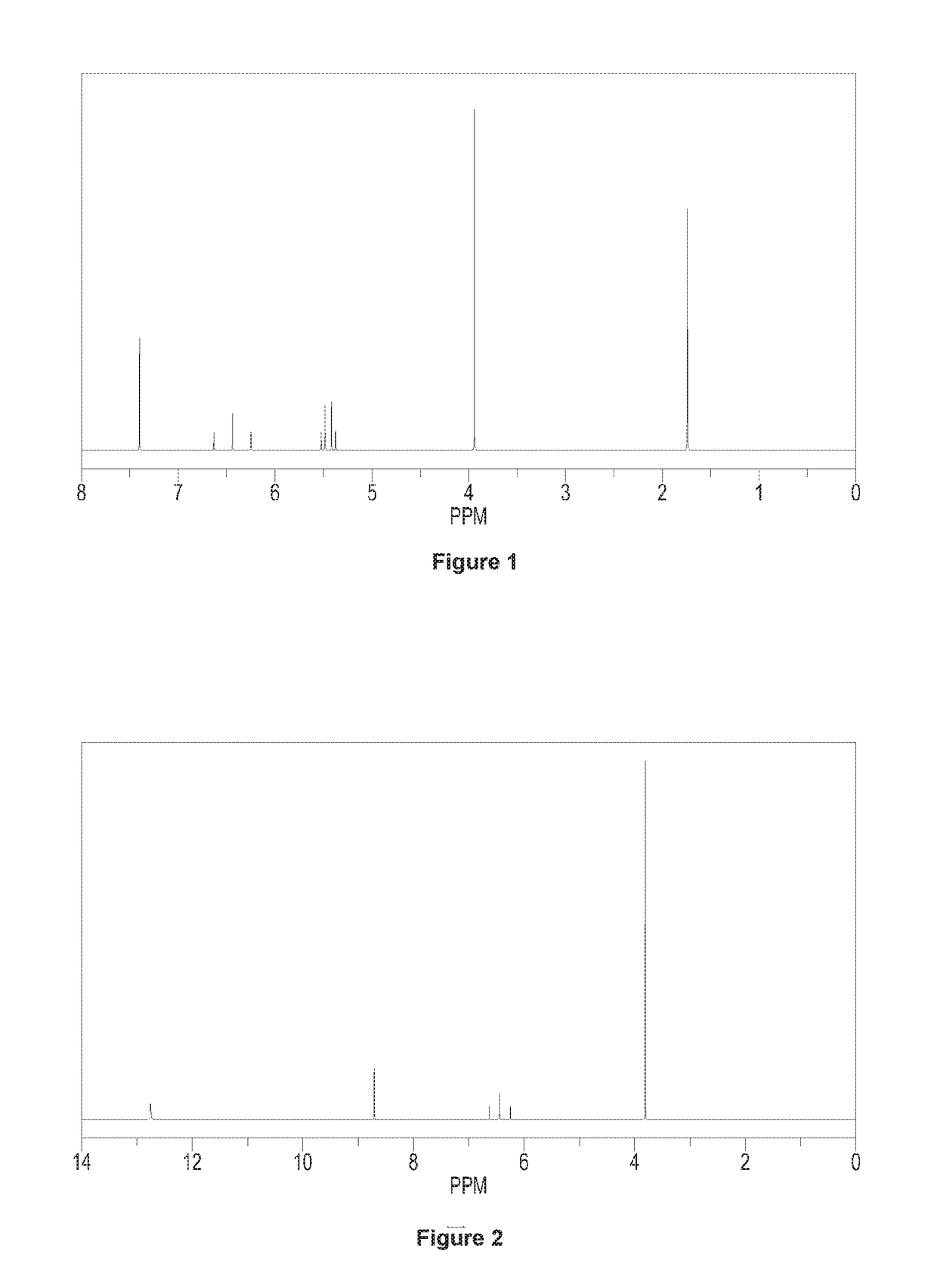
[0091]Adding 70 g N-1,1,2,2-tetrafluoro-ethyl dimethyl amine (0.48 mol) and 300 g acetonitrile to a 1000 ml four orifices reaction bulb equipped with a stirring device and a thermometer, and then dropping 150 g BF3 acetonitrile solution into the reaction bulb at 20° C. (wherein, with respect to the amount of N-1,1,2,2-tetrafluoro-ethyl dimethyl amine, containing 1.2 equivalents of BF3), dropwise over 15-30 min, and stirring the reactants in the reaction bulb for 30 min. Dropping 81.8 g (0.58 mol) N,N-dimethyl-2-(2-methyl-1,3-dioxo-2-yl)-vinyl into the reaction bulb dropwise over 30 min. Rising the temperature of the above reaction system gradually to 20° C., and carrying out the reaction in the bulb with heat insulation for 2 hours. Cooling down the temperature of the reaction system to 5° C. after 2 hours heat insulation, and dropping acetonitrile solution of methyl hydrazine (wherein, with respect to the
example 2
ration Method of 3-(difluoromethyl)-1-methyl-1H-pyrazole-4-carboxylic Acid
[0100]
[0101]Adding 73 g (0.5 mol) N-1,1,2,2-tetrafluoro-ethyl dimethyl amine and 300 g acetonitrile to a 1000 ml four orifices reaction bulb equipped with a stirring device and a thermometer, and then dropping 180 g BF3 acetonitrile solution into the reaction bulb at 0° C. (wherein, with respect to the amount of N-1,1,2,2-tetrafluoro-ethyl dimethyl amine, containing 1.3 equivalents of BF3), dropwise over 15-30 min, and stirring the reactants in the reaction bulb at room temperature for 30 min. Dropping 78 g (0.55 mol) N,N-dimethyl-2-(2-methyl-1,3-dioxo-2-yl)-vinyl into the reaction bulb, dropwise over 30 min. Carrying out the reaction in the bulb at 30° C. with heat insulation for 2 hours. Cooling down the temperature of the reaction system to 5° C. after 2 hours heat insulation, and dropping acetonitrile solution of methyl hydrazine (wherein, with respect to the amount of 1,5-diaza pentadiene salt, containing 1.
example 3
ration Method of 3-(difluoromethyl)-1-methyl-1H-pyrazole-4-carboxylic Acid
[0106]
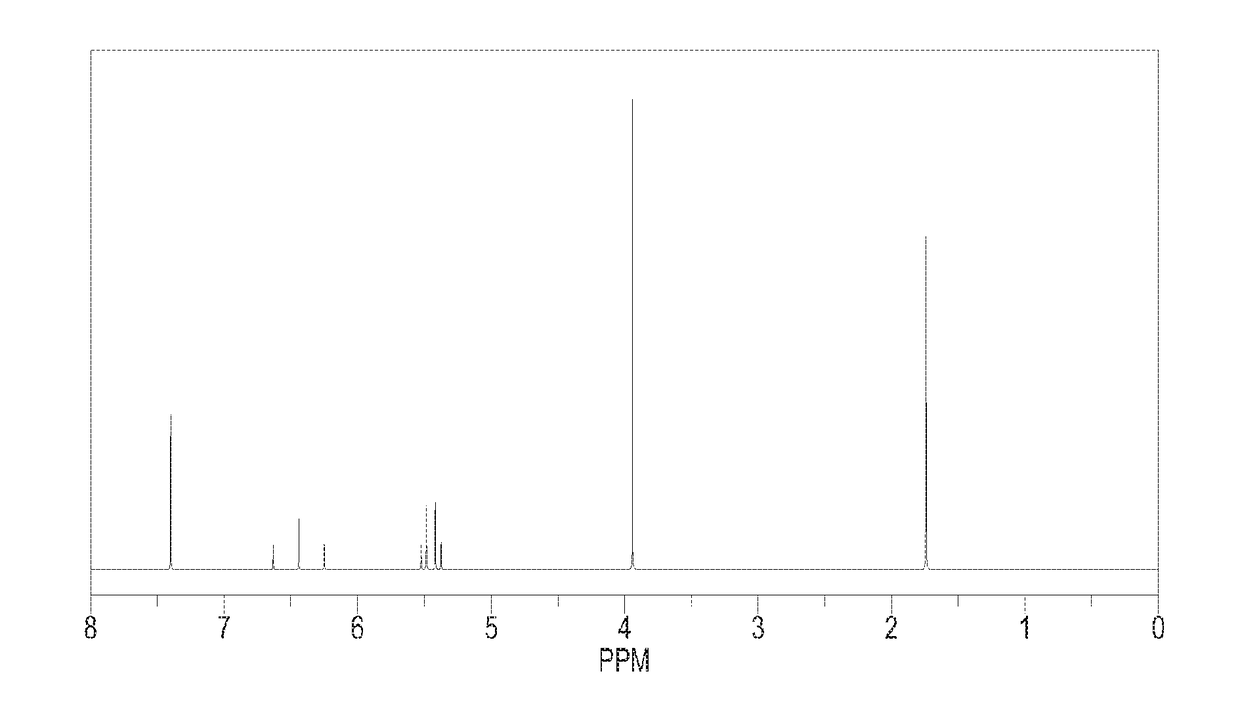
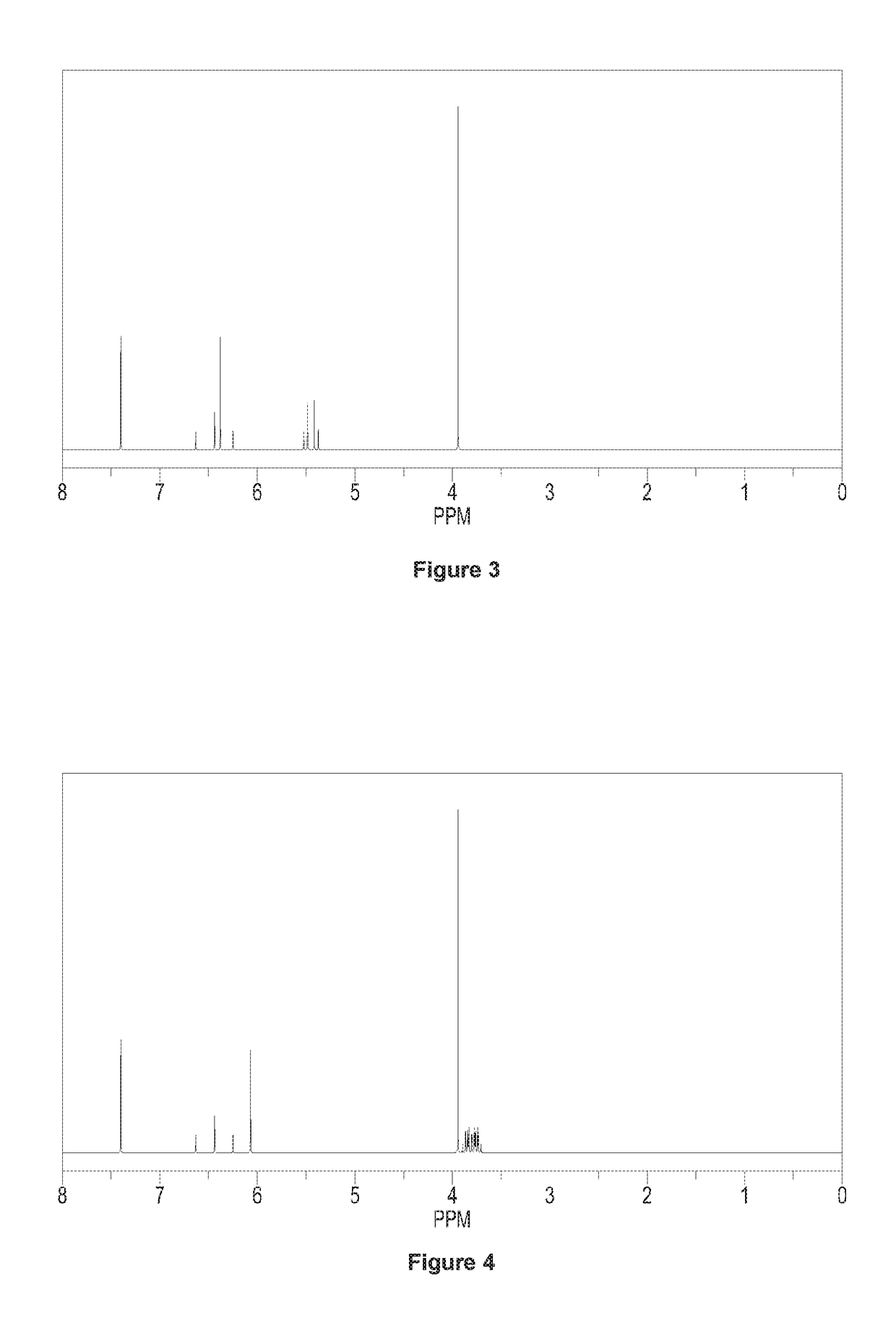
[0107]Except using 0.58 mol N,N-dimethyl-2-(1,3-dioxo-2-yl)-vinyl instead of using N,N-dimethyl-2-(2-methyl-1,3-dioxo-2-yl)-vinyl, repeating the preparation method of 3-(difluoromethyl)-1-methyl-4-(2-methyl-1,3-dioxetan-2-yl)-1H-pyrazole in Example 1. Calculating the molar yield with respect to the mole of N-1,1,2,2-tetrafluoro-ethyl dimethyl amine, said molar yield is 90.0%. Said target product has been analyzed by 1H NMR analysis, elemental analysis and mass spectrometry, which could be determined as 3-(difluoromethyl)-4-(1,3-dioxetan-2-yl)-1-methyl-1H-pyrazole.
[0108]elemental analysis result and mass spectrometry result of said target product are as follows:
[0109]mass spectrometry: m / z: 190.06 (100.0%), 191.06 (7.7%);
[0110]elemental analysis: C, 44.22; H, 4.24; F, 19.98; N, 14.73; O, 16.83.
[0111]Repeating the preparation method of 3-(difluoromethyl)-1-methyl-1H-pyrazole-4-carboxylic acid in Example 1 b
PUM
| Property | Measurement | Unit |
|---|---|---|
| Temperature | aaaaa | aaaaa |
| Temperature | aaaaa | aaaaa |
| Temperature | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap