Medical equipment for platelet-rich plasma gel (PRG) in-vitro negative pressure cutting treatment
A technology for platelet-rich plasma and medical equipment, applied in the field of medical equipment, can solve the problems of sterile level PRP affecting repair quality, lack of efficient and safe cutting equipment, and inability to cut in suitable shape, so as to reduce the demand for autologous plasma. , The effect of retaining PRP activity and reducing the probability of contamination
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
Embodiment
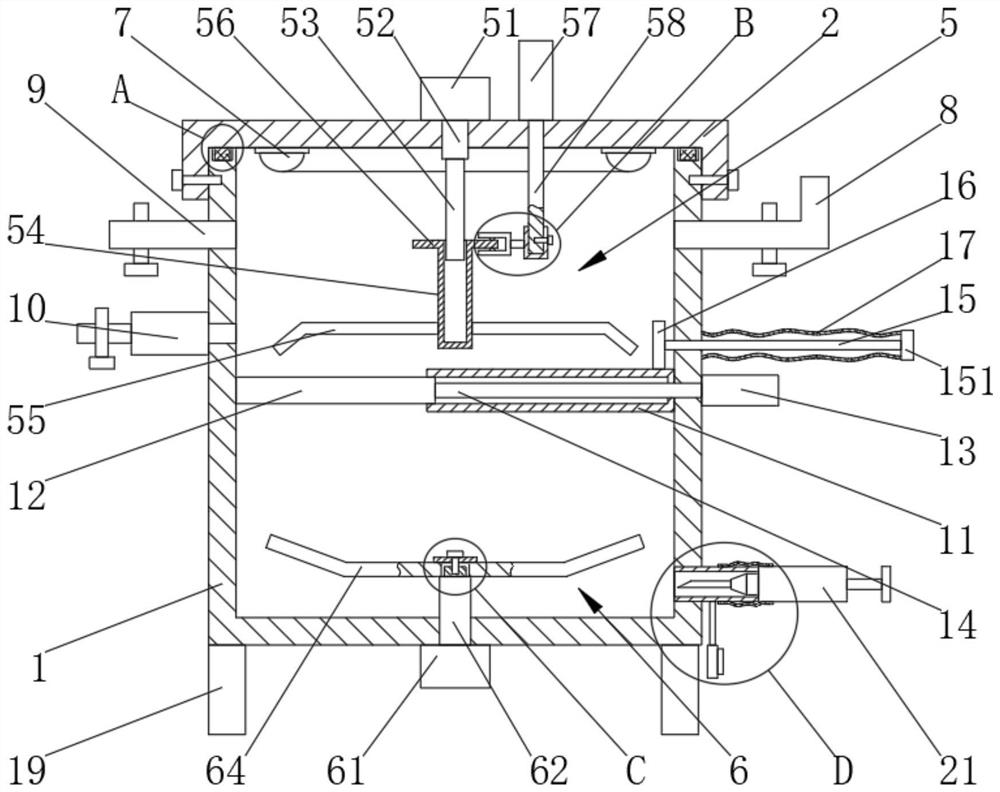
[0026] The medical equipment for in vitro negative pressure cutting of platelet-rich plasma gel PRG includes a box body 1, the box body 1 is connected with a box cover 2 by screws, the box body 1 is provided with a sealing groove 3, and the box body 1 is provided with a sealing groove 3. The box cover 2 is bonded with a silicone ring 4, the sealing groove 3 is slidably connected with a silicone ring 4, the box cover 2 is provided with a lifting and stirring mechanism 5, and the box body 1 is provided with a high-speed cutting mechanism 6, A UV lamp 7 is fixedly installed on the box cover 2, a PRP guide port 8 is fixedly sleeved on the side wall of the box body 1, and an activator guide port 9 is fixed and sleeved on the side wall of the box body 1. The side wall of 1 is fixedly installed with a suction pump 10. The box body 1 is welded with four legs 19. The legs 19 are evenly distributed in the box body 1. The box body 1 is provided with an outlet 20. The outlet 20 A needle cyli
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap