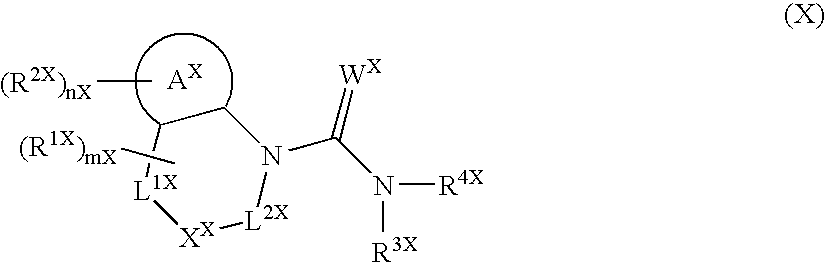
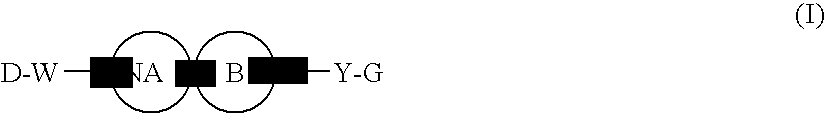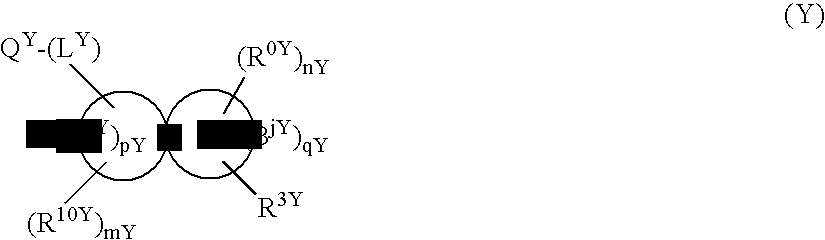Heterocyclic Spiro Compound
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
1-tert-butyl 3-ethyl piperidine-1,3-dicarboxylate
[0303] To a solution of ethyl nipecotinate (15.5 mL, 99.8 mmol) in tetrahydrofuran (100 mL) was added di-tert-butyl dicarbonate (21.8 g, 10.0 mmol) under ice-cooling, and the mixture was stirred for 2 hours at room temperature. The reaction mixture was concentrated. The obtained residue was purified by column chromatography on silica gel (hexane:ethyl acetate=5:1) to give the title compound (25.5 g) having the following physical data.
[0304] TLC: Rf 0.42 (hexane:ethyl acetate=5:1);
[0305] NMR: δ 1.26 (t, J=7.14 Hz, 3H), 1.55 (m, 12H), 2.03 (m, 1H), 2.43 (m, 1H), 2.80 (m, 1H), 2.96 (s, 1H), 3.91 (m, 1H), 4.13 (m, 3H).
example 2
1-tert-butyl 3-ethyl 3-allylpiperidine-1,3-dicarboxylate
[0306] A lithium diisopropylamide (2.0 mol / L in mixture of tetrahydrofuran-heptane-ethylbenzene, 22.5 mL, 45.0 mmol) was added to tetrahydrofuran (50 mL) and the mixture was cooled to −78° C. To the cooled mixture was added dropwise the solution of the compound prepared in Example 1 (7.72 g, 30.0 mmol) in tetrahydrofuran (50 mL) during 30 minutes, and the resulting mixture was stirred for 1 hour. The reaction mixture was added dropwise allyl bromide (3.9 mL, 45.1 mmol) and the mixture was stirred for 1 hour at −78° C. The reaction mixture then was allowed to warm gradually to ambient temperature. The mixture was added 1N hydrochloric acid and then extracted with ethyl acetate. The organic layer was washed with water, a saturated aqueous solution of sodium bicarbonate and a saturated aqueous solution of sodium chloride sequentially, dried over an anhydrous magnesium sulfate and then concentrated. The residue was purified by colu
example 3
1-tert-butyl 3-ethyl 3-(2-oxoethyl)piperidine-1,3-dicarboxylate
[0309] To a solution of the compound prepared in Example 2 (1.01 g, 3.40 mmol) in dichloromethane (17 mL) was added Sudan III (trace) as an indicator and the mixture was cooled to −60° C. Ozone was bubbled through the solution with stirring for about 15 minutes until a pale red color disappeared. Argon was bubbled through the solution for 10 minutes. To the solution was added dropwise dimethylsulfide (10.0 mL, 136 mmol). The mixture was allowed to warm gradually to ambient temperature and then was stirred overnight. The mixture was concentrated and the obtained residue was purified by column chromatography on silica gel (hexane:ethyl acetate=4:1) to give the title compound (784 mg) having the following physical data.
[0310] TLC: Rf 0.33 (hexane:ethyl acetate=3:1);
[0311] NMR: δ 1.25 (t, J=7.14 Hz, 3H), 1.43 (s, 9H), 1.62 (m, 2H), 1.75 (m, 1H), 1.93 (m, 1H), 2.65 (m, 1H), 2.82 (m, 1H), 2.99 (m, 1H), 3.44 (m, 1H) 3.81 (m, 2H
PUM
| Property | Measurement | Unit |
|---|---|---|
| Affinity | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap