Alpha 2 Delta Ligands for Fibromyalgia and Other Disorders
a technology of fibromyalgia and other disorders, which is applied in the direction of immunological disorders, drug compositions, peptide/protein ingredients, etc., can solve the problems of disappointing clinical trials and modest success of treating fibromyalgia with a single pharmacological agent, and achieve the effects of decreasing slow wave sleep, increasing slow wave sleep, and increasing slow wave sleep
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 2
Synthesis of Example 2
(3R,4S)-3-Aminomethyl-4,5-dimethyl-hexanoic acid
[0271]
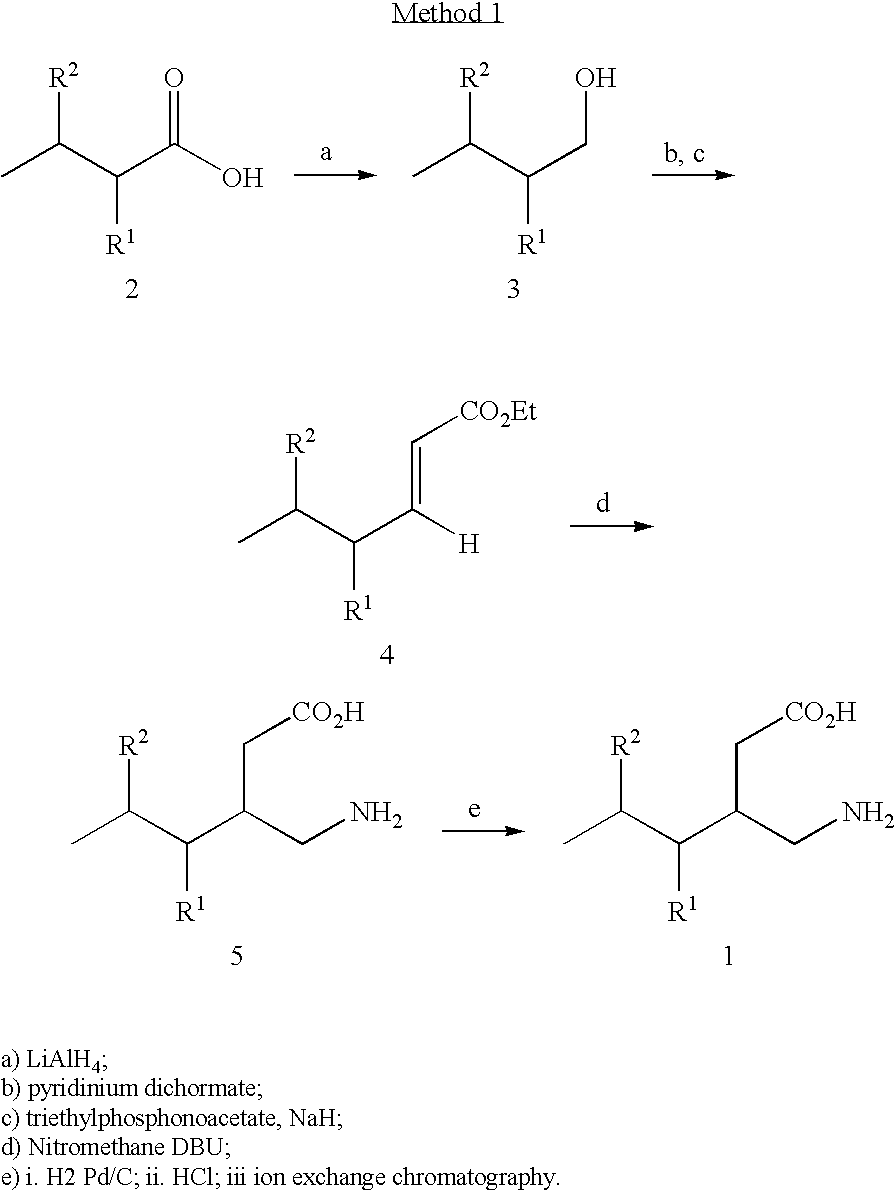
Reagents and Conditions
[0272]a) (R)-(−)-4-phenyl-2-oxazolidinone, (CH3)3CCOCl, Et3N, LiCl, THF, −20 to 23° C.;[0273]b) MeMgCl, CuBrSMe2, THF, −35° C.;[0274]c) NaHMDS, BrCH2CO2tBu, THF, −78° C. to −40° C.;[0275]d) LiOH, H2O2, THF, H2O, 25° C.;[0276]e) BH3SMe2, THF, 0 to 25° C.;[0277]f) pTsCl, pyridine, 25° C.;[0278]g) NaN3, DMSO, 60° C.;[0279]h) Raney nickel, MeOH, H2; i) 3M HCl, reflux, ion exchange resin (Dowex 50WX8, strongly acidic).
[R-(E)]3-(4-Methyl-pent-2-enoyl)-4-phenyl-oxazolidin-2-one 16
[0280]Trimethylacetyl chloride (7.8 g, 0.065 mol) was added to acid 14 (6.9 g, 0.06 mol) and triethylamine (18 g, 0.187 mol) in THF (200 mL) at −20° C. After 1 hour, lithium chloride (2.35 g, 0.55 mol) and (R)-(−)-4-phenyl-2-oxazolidinone (8.15 g, 0.05 mol) were added and the thick suspension warmed to room temperature. After 20 hours, the suspension was filtered and the filtrate concentrated. The resultant solid was re
specific examples
Example 3
Synthesis of 3-Aminomethyl-5-methyl-octanoic acid
[0635]
1-Benzyl-4-hydroxymethyl-pyrrolidine-2-one 74
[0636]Sodium borohydride (8.0 g, 0.211 mol) was added to a solution of methyl-1-benzyl-5-oxo-3-pyrrolidnecarboxylate 73 (See Zoretic et al, J. Org. Chem., 1980; 45:810-814 for general method of synthesis) (32.0 g, 0.137 mol) in 1,2-dimethoxyethane (600 mL) and refluxed for 19 hours. The reaction was cooled to room temperature and 200 mL of water was added. The reaction was quenched with 1 M citric acid and concentrated under reduced pressure. The residue was extracted with dichloromethane, dried over magnesium sulfate, and evaporated to dryness to give 17.47 g, 62% of the alcohol 74 as clear oil. 1H NMR (CDCl3) δ 7.30 (m, 5M), 4.38 (d, 1H, J=14.7), 4.46 (d, 1H, J=14.7), 3.56 (m, 2H), 3.36 (m, 1H), 3.10 (m, 1H), 2.52 (m, 2H), 2.26 (m, 1H). MS, m / z (relative intensity): 207 [M+2H, 66%]. IR (KBr) 3345, 2946, 2866, 1651, 1445, 1025, 737, and 698 cm−1.
1-Benzyl-4-iodomethyl-pyrrolid
example 4
Synthesis of 3-Aminomethyl-5,7-dimethyl-octanoic acid
[0641]
1-(4-Methoxy-benzyl)-5-oxo-pyrrolidine-3-carboxylic acid methyl ester 79
[0642]To 4-methoxybenzylamine (42 g, 0.306 mol) in methanol (40 mL) at 0° C. was added the dimethyl itaconate (48 g, 0.306 mol) in methanol (13 mL). The solution was stirred at room temperature for 4 days. 1N HCl was added to the solution followed by ether. The two layers were separated and the aqueous phase extracted with ether. The combined organic phases were dried (MgSO4). Upon filtration of the drying agent the desired material 79 precipitated from solution that was collected and dried under vacuum. 23.26 g, 29%. MS, m / z (relative intensity): 264 [M+H, 100%]. Anal. Calcd for C14H17N1O4: C, 63.87; H, 6.51; N, 5.32. Found: C, 63.96; H, 6.55; N, 5.29.
4-Hydroxymethyl-1-(4-methoxy-benzyl)-pyrrolidine-2-one 80
[0643]NaBH4 (15 g, 0.081 mol) was added in portions to ester 79 in ethanol (600 mL) at room temperature. After 4.5 hours water (˜200 mL) was carefull
PUM
| Property | Measurement | Unit |
|---|---|---|
| Electrical conductance | aaaaa | aaaaa |
Abstract
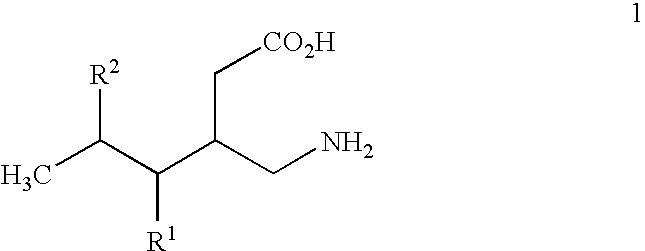
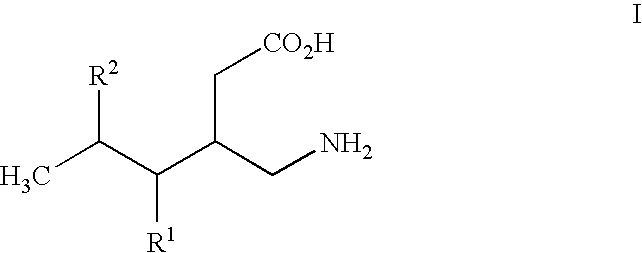
- R1 is hydrogen, straight or branched alkyl of from 1 to 6 carbon atoms or phenyl; and
- R2 is straight or branched alkyl of from 4 to 8 carbon atoms, straight or branched alkenyl of from 2 to 8 carbon atoms, cycloalkyl of from 3 to 7 carbon atoms, alkoxy of from 1 to 6 carbon atoms, -alkylcycloalkyl, -alkylalkoxy, -alkyl OH, -alkylphenyl, -alkylphenoxy, or -substituted phenyl. The invention also relates to a method of treating the above disorders by administering the compound (3S,5R)-3-aminomethyl-5-methyl-octanoic acid.
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap