Dengue virus degeneration vaccine and application thereof
A dengue virus and vaccine technology, applied in antiviral immunoglobulins, applications, viral peptides, etc., can solve the problem that the type 2 dengue virus strain cannot play a protective role, the protection effect of specific epidemic strains is poor, and the protective power is relatively different. Large and other problems, to achieve the effects of low mammalian cell expression system, cost control, and improved safety of use
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Example Embodiment
[0044] Example 1 Design of Dengue Virus Degenerate Sequence Vaccine Molecule
[0045] 1. Sequence alignment
[0046] DV is widespread in tropical and subtropical regions, especially in Southeast Asia, Africa, Central and South America and the Western Pacific. The present invention selects DV strains that have been popular in the world, and the DV strains include representative strains of four serotypes: DV-1, DV-2, DV-3 and DV-4. DV-1 selected epidemic strains in Southwestern Indian Ocean and Africa (GenBank accession number DQ285558), Indonesia epidemic strain (GenBank accession number AB189121), American epidemic strain (GenBank accession number GQ868530) and China epidemic strain (GenBank accession number AY376738); DV- 2 Select the TR1751 strain popular in the United States and Japan (Trent, et al.1983), the western African strain (GenBank accession numbers EF105386 and EF105378), the American epidemic strain (GenBank accession number GQ199892) and the Pacific region epidemic s
Example Embodiment
[0063] Example 2 Construction of recombinant expression plasmid of dengue virus degenerate vaccine
[0064] 1. The chemically synthesized degenerate sequence DVIII is double digested with BamHI (TaKaRa company) and XhoI (TaKaRa company)
[0065]
[0066] After mixing, place it in a water bath at 37°C for 4 hours, perform electrophoresis on 1.2% agarose (Shanghai Shenggong) gel, and recover the double digested DVIII fragment according to the instructions of the Promega gel recovery kit;
[0067] 2. The prokaryotic expression vector pET22b was double digested with BamHI (TaKaRa company) and XhoI (TaKaRa company). The double digestion reaction system was the same as above, and the plasmid was recovered;
[0068]
[0069] After mixing, put it in a 16℃ water bath for 16 hours, and then inactivate the ligase at 65℃ for 10 minutes;
[0070] 4. Conversion experiment
[0071] For the preparation of Escherichia coli DH5α competence, please refer to the Molecular Cloning Experiment Guide (third edit
Example Embodiment
[0072] Example 3 Screen and identify recombinant bacterial clones
[0073] 1. The recombinant expression plasmid pET22b-DVIII described in Example 2 was transformed into BL21 Escherichia coli competent according to the method of Molecular Cloning Experiment Guide (third edition, Science Press, 2002), please refer to the description in step 4 in Example 2 for details Method to proceed.
[0074] 2. Pick 10 AMP resistant clones and culture them with 1ml LB medium. When the OD600 value is about 0.3-0.5, add 0.5mM IPTG (Beijing Saibaisheng) inducer for 1 hour and centrifuge at 14,000 rpm for 5 Minutes, collect the bacteria.
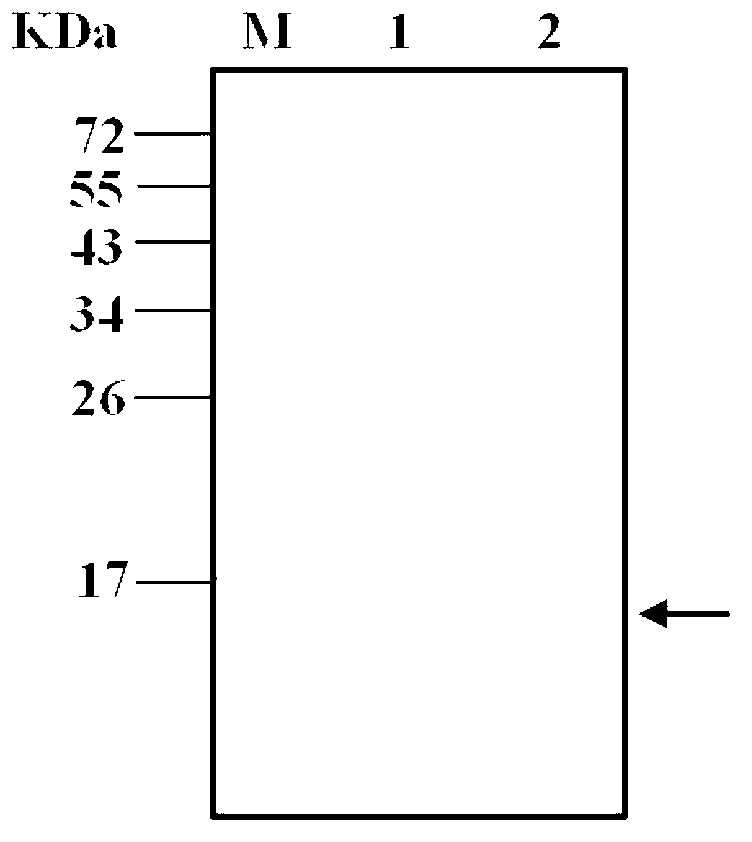
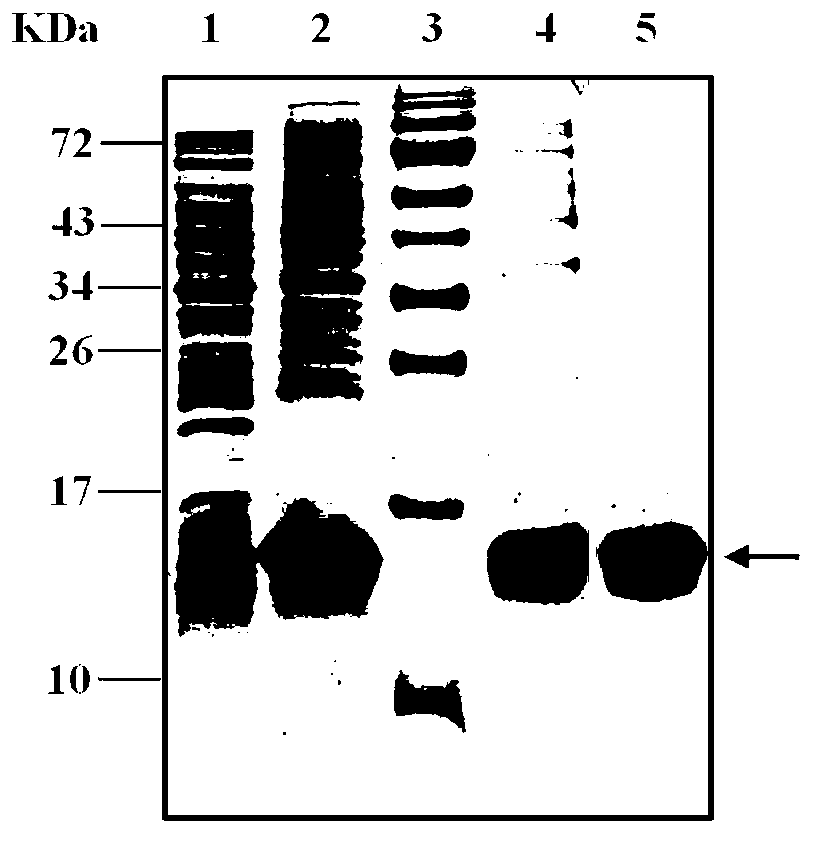
[0075] 3. Carry out SDS-PAGE electrophoresis (Bio-Rad, USA) according to the method of molecular cloning experiment guide. After electrophoresis, the gel is stained with Coomassie blue (Fluka, Switzerland). Observe that compared with the uninduced strain, there is The clone that clearly expressed the band was a positive expression clone, named pET22b-DVIII / BL21 en
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap