Composite adjuvant and novel coronavirus COVID-19 recombinant protein vaccine using composite adjuvant
A COVID-19, recombinant protein technology, applied in the field of new crown COVID-19 recombinant protein vaccine, can solve the problems of limited immune enhancement, pain, allergies, etc., and achieve the effect of rapid preparation
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Example Embodiment
[0041] Example 1
[0042] This embodiment provides a composite adjuvant composed of CpG oligodeoxynucleotide and aluminum hydroxide; wherein, the mass ratio of CpG oligodeoxynucleotide and aluminum hydroxide is 1:0.6.
[0043] This embodiment further provides a combination of the compound adjuvant and the novel coronavirus COVID-19 recombinant vaccine.
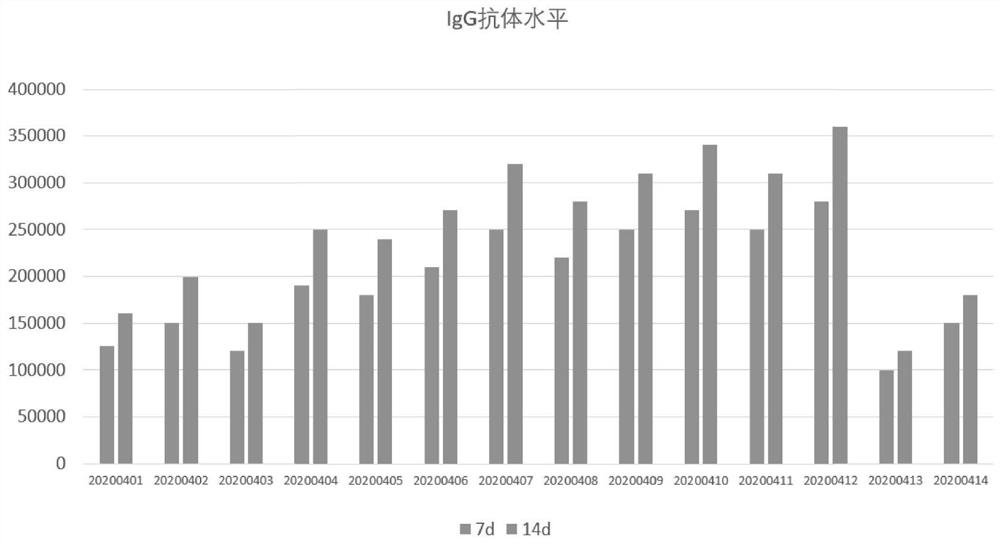
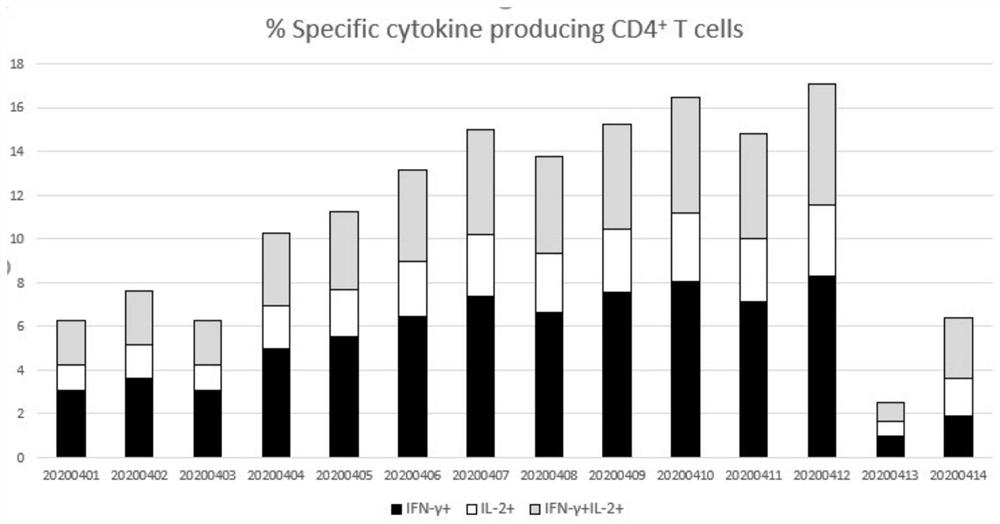
[0044] Specifically: add CpG oligodeoxynucleotide adjuvant and aluminum hydroxide adjuvant to the vaccine stock solution filtered by a 0.22 μm filter membrane, so that the concentration of CpG adjuvant is 0.5 mg / dose and the content of aluminum hydroxide adjuvant is 0.3 mg / dose, vaccine antigen content 20μg / dose. The vaccine number obtained in this example is recorded as 20200401.
Example Embodiment
[0045] Example 2
[0046] This embodiment provides a composite adjuvant composed of CpG oligodeoxynucleotide and aluminum hydroxide; wherein, the mass ratio of CpG oligodeoxynucleotide and aluminum hydroxide is 1:0.6.
[0047] This embodiment further provides a combination of the compound adjuvant and the novel coronavirus COVID-19 recombinant vaccine.
[0048] Specifically: add CpG oligodeoxynucleotide adjuvant and aluminum hydroxide adjuvant to the vaccine stock solution filtered by a 0.22 μm filter membrane, so that the concentration of CpG adjuvant is 0.5 mg / dose and the content of aluminum hydroxide adjuvant is 0.3 mg / dose, vaccine antigen content 40μg / dose. The vaccine number obtained in this example is recorded as 20200402.
Example Embodiment
[0049] Example 3
[0050] This embodiment provides a composite adjuvant composed of CpG oligodeoxynucleotide and aluminum hydroxide; wherein, the mass ratio of CpG oligodeoxynucleotide and aluminum hydroxide is 1:0.9.
[0051] This embodiment further provides a combination of the compound adjuvant and the novel coronavirus COVID-19 recombinant vaccine.
[0052] Specifically: add CpG oligodeoxynucleotide adjuvant and aluminum hydroxide adjuvant to the vaccine stock solution filtered by 0.22 μm filter membrane, so that the concentration of CpG adjuvant is 0.5mg / dose, and the content of aluminum hydroxide adjuvant is 0.45mg / dose, vaccine antigen content 20μg / dose. The vaccine number obtained in this example is recorded as 20200403.
PUM
| Property | Measurement | Unit |
|---|---|---|
| Chain length | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap