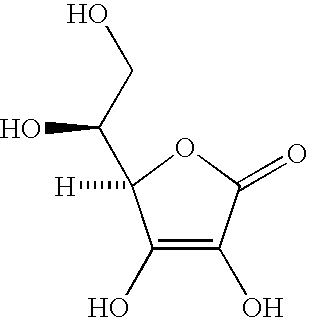
Stable ascorbic acid compositions
a technology of ascorbic acid and composition, applied in the field of ascorbic acid composition, can solve the problems of poor wound healing, edema, scurvy, and hemorrhage under the skin, and achieve the effects of improving stability, excellent stability, and satisfactory shelf li
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
[0041] Example 1 below shows a non-limiting example of a suitable composition in accordance with the present disclosure.
Weight percent of the totalIngredientcompositionWater60-94% Sorbitol 70%0.1-10% Ca Hydroxide0.10-0.5% Zn Chloride0.10-2% Na Hyalurate0.001-0.02% Ascorbic acid 5-40%SLS (30% solution) 0-5%Phenoxyethanol0.1-1%Fragrance0.0-5%Alkydimethylbenzylamine 0-5%
example 2
[0042] Example 2 below shows another suitable non-limiting example of a composition in accordance with the present disclosure.
Weight percent of the totalIngredientcompositionWater60-94%Reducing Sugar0.1-10% Metal Salt or mixtures thereof0.10-5% Moisturizing Agent0.0-0.02% Ascorbic acid 5-40%Antimicrobial0.0-1%Surfactant0.0-5%Fragrance0.0-5%Alcohol0.0-20%
[0043] The compositions of the present disclosure may be packaged in suitable containers such as tubes or bottles. Suitable containers are commercially available from a variety of suppliers. A wide variety of containers and suppliers are listed in the CPC Packaging Directory. (See, Buyers' Guide under “Containers” at www.cpcpkg.com). In embodiments, containers are selected with low oxygen permeability. Suitable containers include containers made from high density polyethylene and the like.
example 3
Stability Study
[0044] In vitamin C compositions without reducing sugar, an aqueous solution of 5% ascorbic acid will likely decompose to less than 90% of the concentration at room temperature in 4 weeks time. See, for example, U.S. Pat. No. 4,983,382 the entire disclosure of which is incorporated herein by this reference.
[0045] Conversely, the stability of compositions made in accordance with the present disclosure show improved stability. Such compositions were evaluated by placing aliquots of each example in an oven at 5, 25, 30 and 40 degrees Centigrade for predetermined time periods and at the end of each time period analyzing the amount of vitamin C present in the composition.
[0046] The following results were observed with compositions in accordance with example 1 having 20% initial vitamin C concentration, sorbitol 70%, Ca hydroxide, Zn chloride, Na hyaluronate 1%, SLS (30% solution), phenoxyethanol and fragrance.
Stability of formulation of example No. 1% Vitamin CInitial a
PUM
| Property | Measurement | Unit |
|---|---|---|
| Fraction | aaaaa | aaaaa |
| Fraction | aaaaa | aaaaa |
| Fraction | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap