Immunoadsorption blood purification material and preparation method thereof
An immunoadsorption material and immunoadsorption technology, which are applied in the field of immunoadsorption blood purification materials and their preparation, can solve the problems that the coupled ligands are easy to fall off and enter the human body, the method is complicated, and there are many reaction steps, so as to reduce the risk of bleeding in patients, The preparation process is simple and the adsorption effect is enhanced
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
preparation example Construction
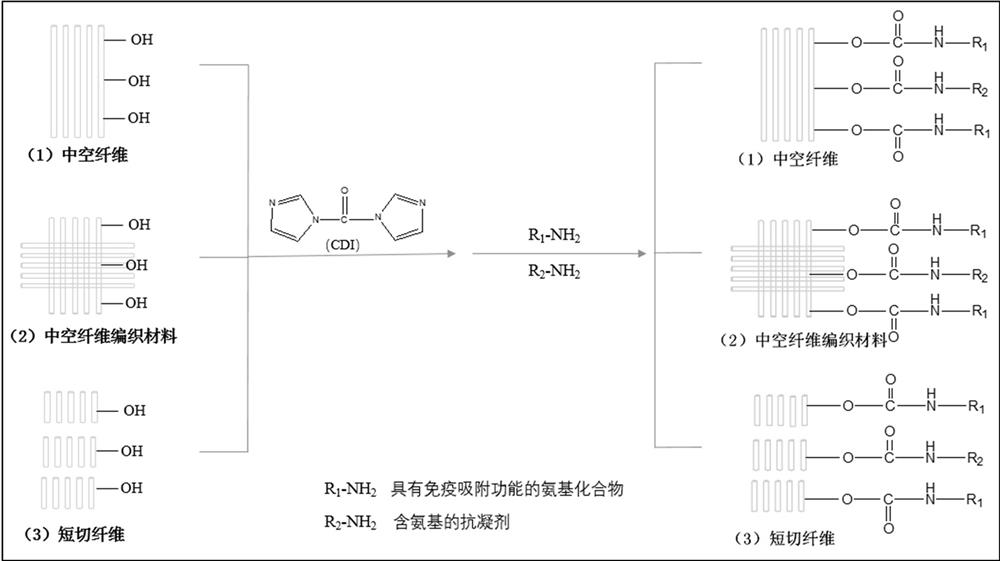
[0054] A method for preparing an immunoadsorbent material, comprising the steps of:
[0055] S1) dissolving carbonyldiimidazole CDI with an organic solvent to obtain a CDI activation solution;
[0056] S2) fully contacting the hydroxyl-containing hollow fiber solid-phase carrier with the CDI activation solution to activate the solid-phase carrier;
[0057] S3) mixing the activated solid-phase carrier with the ligand solution, performing a covalent coupling reaction, and performing post-treatment after the reaction to obtain an immunosorbent material;
[0058] The ligand is an amino compound.
[0059] If the solid phase support itself does not contain hydroxyl groups, it is necessary to use existing techniques, such as using chemical methods to first introduce hydroxyl groups.
[0060] In a specific example, the temperature of the activation reaction is 4°C-25°C. Understandably, in the present invention, the activation reaction temperature includes but not limited to 4°C, 10°C,
Embodiment 1
[0097] This embodiment provides a protein A immunoadsorption material using recombinant protein A and nafamostat mesylate as ligands, specifically as follows:
[0098] S1) Put 100g of dry EVOH hollow fiber (tubular, inner diameter 200μm), 10g of carbonyldiimidazole (CDI) and 10mL of dry acetone in a container, soak and react for 1h at 25°C, take it out and use 70% acetone and dry acetone in sequence Wash to remove imidazole, obtain the hollow fiber matrix containing imidazolyl formate after activation, store the matrix in dry acetone for future use, and mark it as A0;
[0099] S2) Take 100g of A0 prepared above, add 150mL of 0.1mol / L borate buffer solution, control the pH of the system to 7.5-8.5, add 14g of recombinant protein A, 7.5g of nafamostat mesylate, 37°C Soak for 24 hours, stop the reaction, and then wash with 20 times the volume of water for injection;
[0100] S3) After rinsing, add 200mL, 0.2mol / L ethanolamine solution to block the unreacted imidazolyl formate g
Embodiment 2
[0103] This embodiment provides a protein A immunoadsorption material using recombinant protein A and nafamostat mesylate as ligands, specifically as follows:
[0104] S1) Put 100g of dry EVOH hollow fiber (tubular, inner diameter 200μm), 10g of carbonyldiimidazole (CDI) and 10mL of dry acetone in a container, soak and react for 1h at 25°C, take it out and use 70% acetone and dry acetone in sequence washing to remove imidazole, to obtain a hollow fiber matrix containing imidazolyl formate after activation, the matrix is stored in dry acetone for future use, and marked as A0';
[0105] S2) Take 100g of A0' prepared above, add 150mL of 0.1mol / L borate buffer solution, control the pH value of the system to 7.5-8.5, add 7.5g of recombinant protein A, 7.5g of nafamostat mesylate, Soak and react at 37°C for 24 hours, after stopping the reaction, wash with 20 times the volume of water for injection;
[0106] S3) After rinsing, add 200mL, 0.2mol / L ethanolamine solution to block th
PUM
| Property | Measurement | Unit |
|---|---|---|
| The inside diameter of | aaaaa | aaaaa |
| Wall thickness | aaaaa | aaaaa |
| Concentration | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap