Tadalafil crystallization method
A tadalafil and crystallization technology, applied in the field of tadalafil crystallization, can solve problems such as non-compliance with the requirements of the pharmacopoeia, large residues of dimethyl sulfoxide, etc., achieve significant economic value and environmental protection value, reduce energy consumption, The effect of convenient recycling
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0021] Reference Example 1: Preparation of Tadalafil Crude Product
[0022] Referring to patent WO2004011463, crude tadalafil was prepared, purity (HPLC): 99.38%, number of impurities (HPLC): 4, maximum impurity (HPLC): 0.3%.
[0023] Tadalafil HPLC detection method is EP related substance method:
[0024] Chromatographic conditions:
[0025]
[0026]
[0027] Example 1
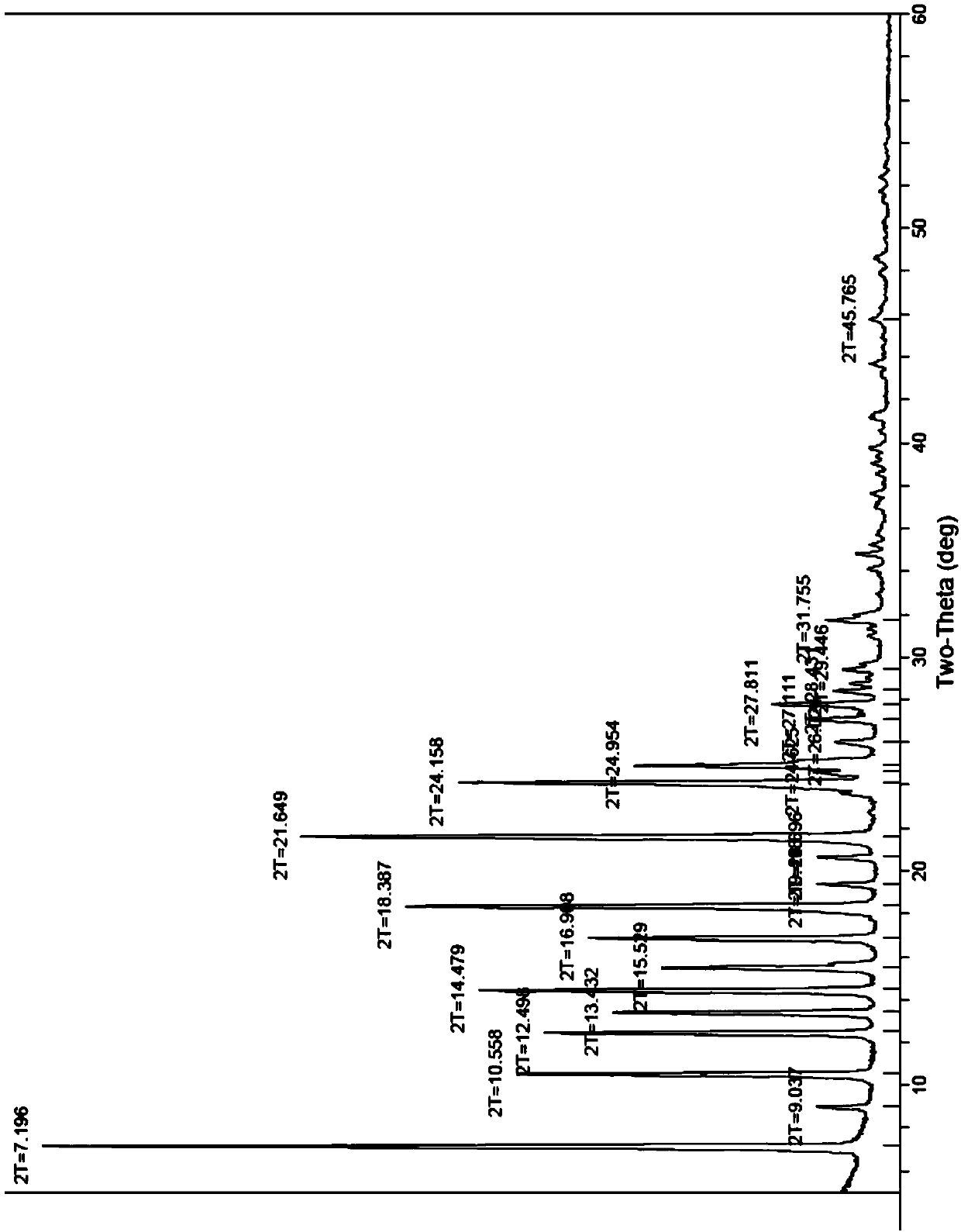
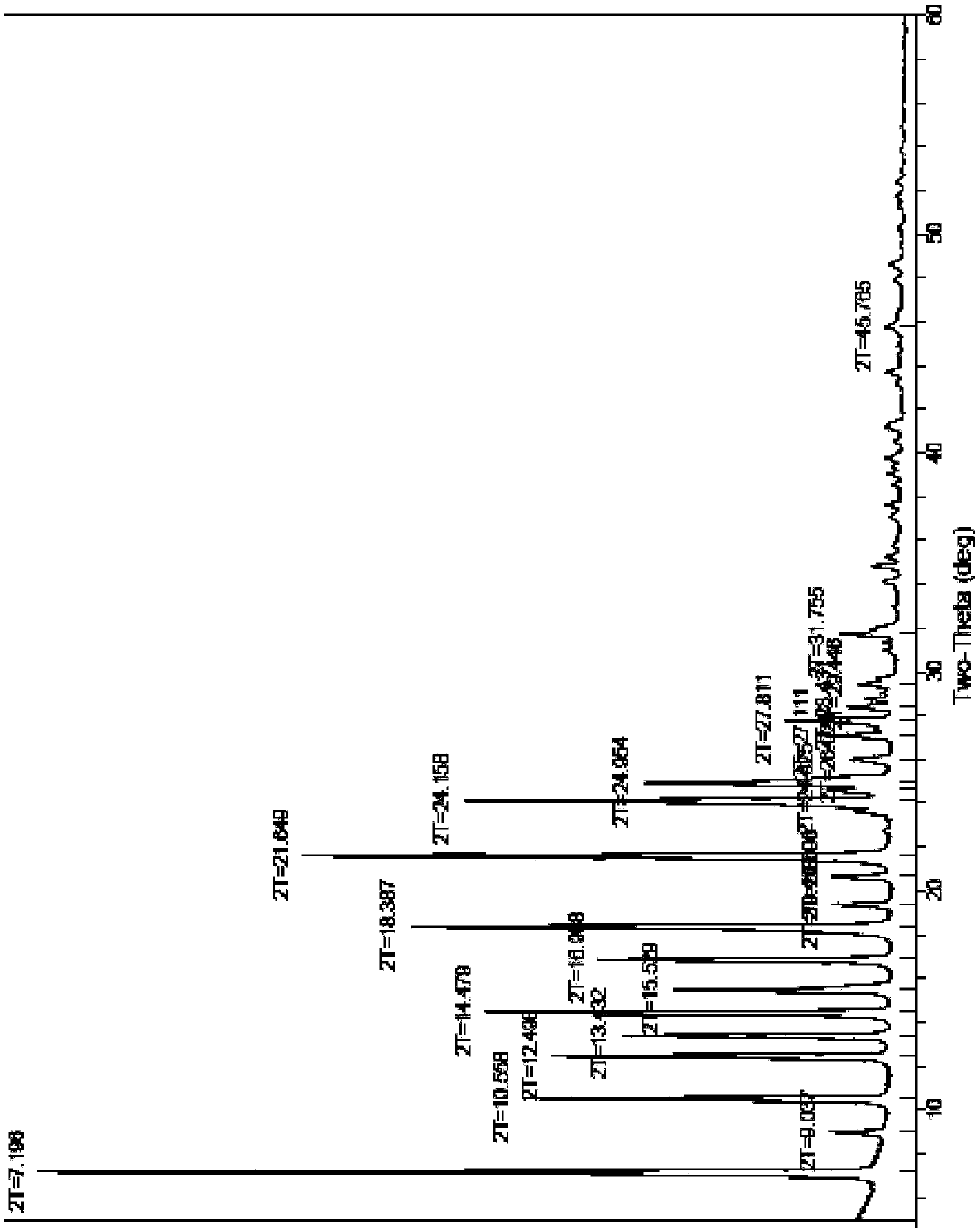
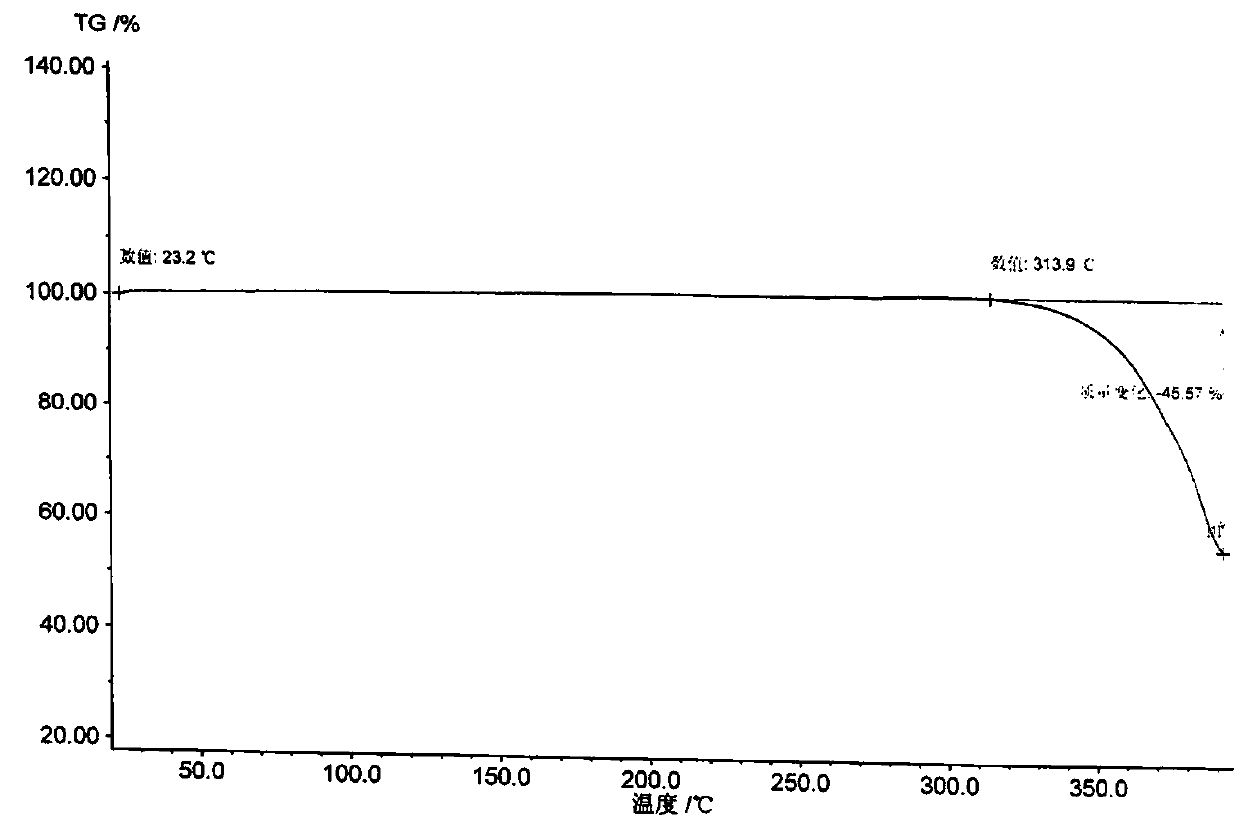
[0028] Get the tadalafil crude product 10g that reference example 1 makes, add 40g dimethyl sulfoxide, stir at room temperature 25 ℃ for 0.5 hour, dissolve clear. Then the dissolved tadalafil crude product solution was added dropwise to 100 g of water under stirring, and the dripping was completed within 30 to 40 minutes. After dropping, continue stirring for 1 hour, filter, wash the wet product with about 20 g of water, and dry it in vacuum at 70°C for 6 hours to obtain 9.0 g of tadala amorphous form A, yield 90%, purity (HPLC): 99.96%, Number of impurities (HPLC): 1, maximum single impurity (HPLC): 0
Embodiment 2
[0031] Take 10 g of the crude tadalafil obtained in Reference Example 1, add 20 g of dimethyl sulfoxide, stir and heat up to 60-70° C., and dissolve. Then, add the dissolved crude tadalafil solution dropwise into 150 g of water under stirring, at room temperature, and control the dropping for 30 to 40 minutes. After dropping, continue stirring at room temperature for 1 hour, filter, wash the wet product with about 20 g of water, and dry it in vacuum at 70°C for 6 hours to obtain 9.5 g of tadalamorph A, yield 95%, purity (HPLC): 99.95 %, number of impurities (HPLC): 2, maximum single impurity (HPLC): 0.03%. Dimethyl sulfoxide residue: 0.011% (pharmacopoeia standard: no more than 0.5%).
Embodiment 3
[0033] Take 10 g of the crude tadalafil obtained in Reference Example 1, add 30 g of dimethyl sulfoxide, stir and heat up to 40° C., and dissolve. Then, add the dissolved crude tadalafil solution dropwise into 200 g of water preheated to 50° C. while stirring, and control the dropping for 30 to 40 minutes. After dropping, continue stirring at room temperature for 1 hour, filter, wash the wet product with about 20 g of water, and dry it in vacuum at 70°C for 6 hours to obtain 9.3 g of tadalamorph A, yield 93%, purity (HPLC): 99.98 %, number of impurities (HPLC): 1, maximum single impurity (HPLC): 0.02%. Dimethyl sulfoxide residue: 0.009% (pharmacopoeia standard: no more than 0.5%).
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap