Administration method for anticancer drugs
a technology of anticancer drugs and administration methods, which is applied in the direction of medical preparations, biological testing, peptides, etc., can solve the problems of destroying cancer cells, affecting the survival rate of cancer cells, and sometimes undergoing apoptosis of cells, so as to reduce side effects of using anticancer drugs and reduce the effect of damag
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example
1. Preparation of Material and Experiments
(1) Cell Culture, Bio-Cycle Synchronization, UV Radiation and Transduction
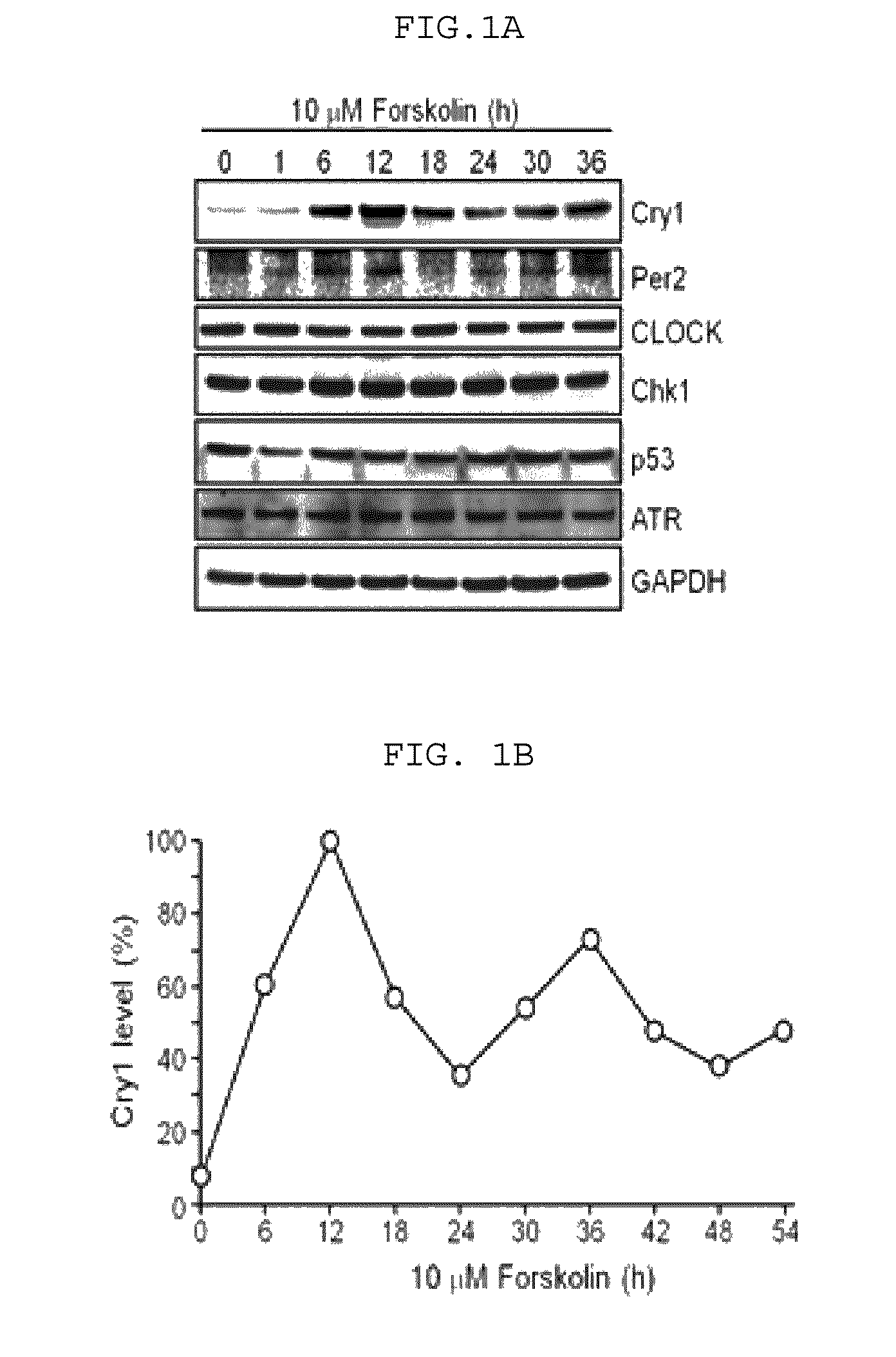
[0134]For cell culture, 1 ml of 100 (vol / vol) fetal bovine serum (FBS, Hyclone Co.) was added to 10 ml of Dulbecco's modified Eagle's medium (DMEM), followed by adding 100 μl of 10 (vol / vol) penicillin-streptomycin thereto. Then, NIH3T3 cells, wild-type mouse embryo fibroblasts (WT MEFs), and mouse embryo fibroblasts free from cryptochrome 1 (Cry1) and cryptochrome 2(Cry2) (CryDKO MEFs), respectively, were cultured in the above prepared culture medium.
[0135]For bio-cycle synchronization, fused cells were further cultured for additional three (3) days after adding 10 mM forskolin to the above medium.
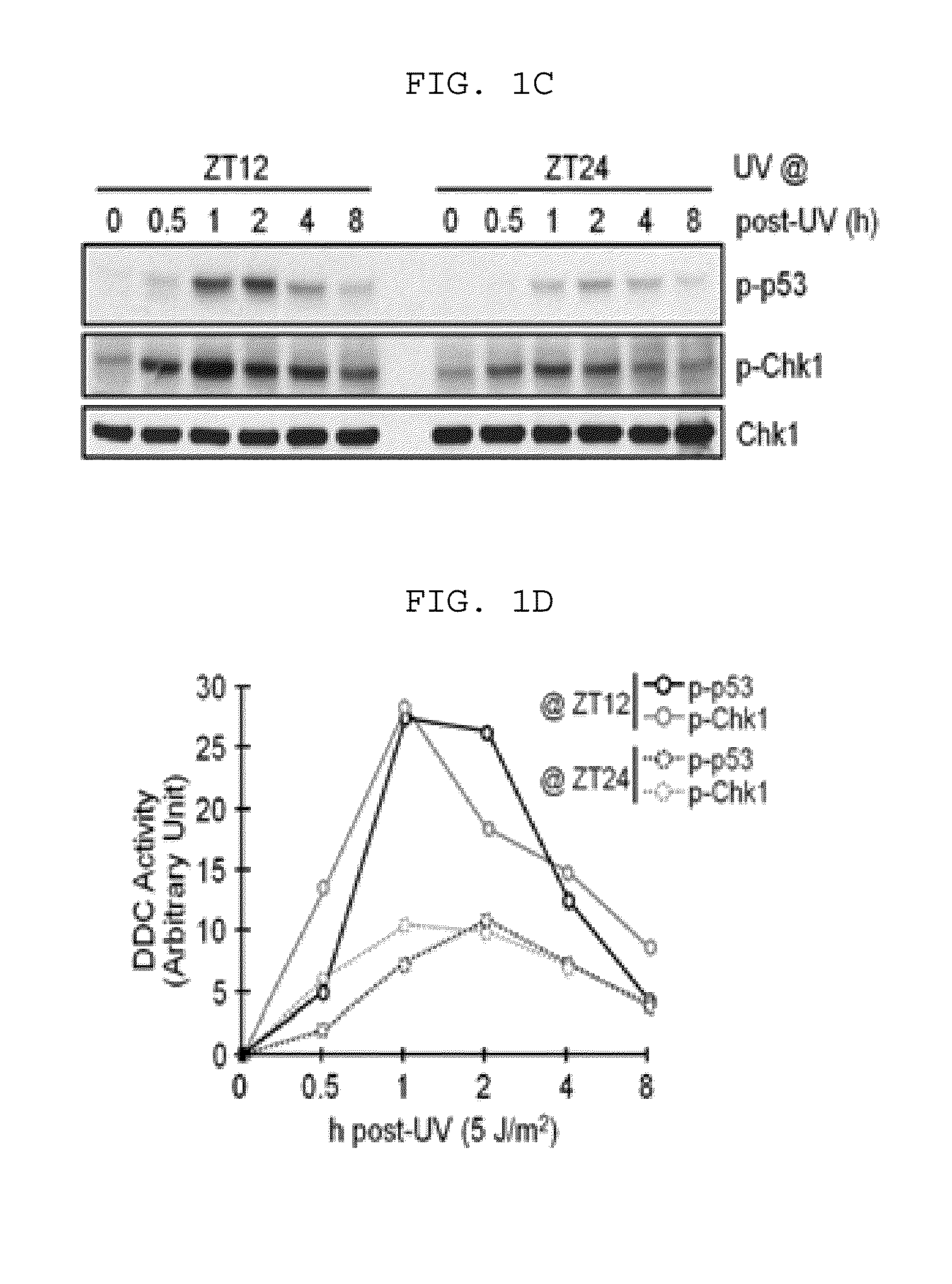
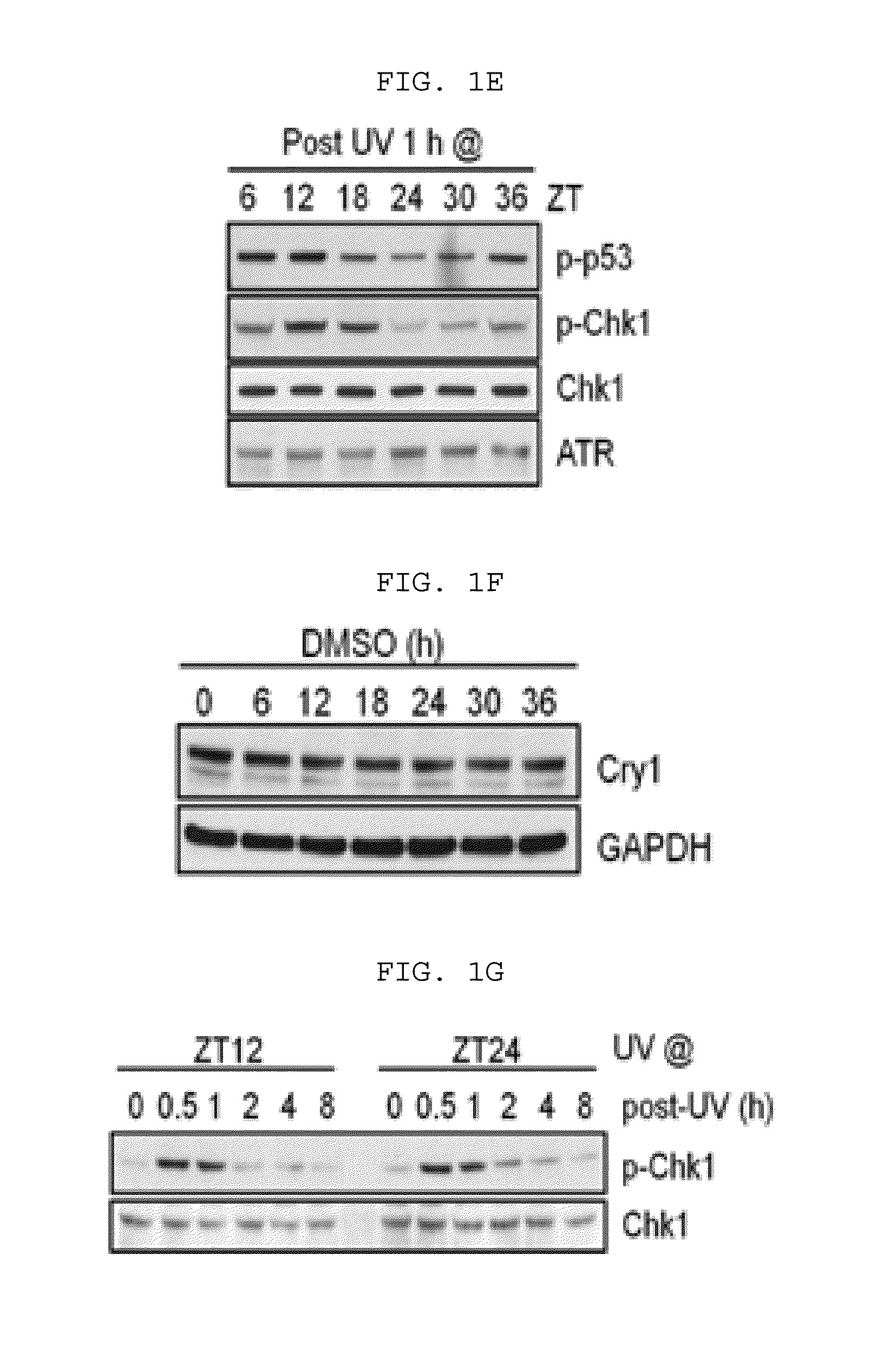
[0136]For UV radiation, using a germicidal lamp (GE) mainly emitting UV-C beam, UV light was emitted to the fused cells by 5 J / m2. Then, a fluorescence rate of the incident beam was measured using a UV-C sensor.
[0137]For transduction of DNA constructs and ON-TARGET plus S
PUM
| Property | Measurement | Unit |
|---|---|---|
| Time | aaaaa | aaaaa |
| Time | aaaaa | aaaaa |
| Time | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap