Method For Preparation of Quick Dissolving Thin Films Containing Bioactive Material With Enhanced Thermal Stability
a bioactive material and thermal stability technology, applied in the field of oral thin films, can solve the problems of large loss of potency, complex products such as over-the-counter medications, dental care and flu medicine, and the process to create oral thin films is generally not designed to encase the large bioactive material, and achieves stable and efficient delivery system, enhanced stability in manufacturing process and storage, and high process recovery
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
esting of OTF's by Fluorescence Focus Assay (FFA)
[0098]A monolayer of confluent MA-104 cells (derived from rhesus monkey kidney tissue obtained from American Type Culture Collection, Manassas, Va.) were grown in 96-well plates for 3-4 days in a medium supplemented with 10% Fetal Bovine Serum (FBS) and kept in a humidified incubator at 37° C., 5% CO2. The old media was replaced with fresh media before infection with the virus. The sterile Oral Thin Film virus sample was transferred into a 10 mL sterile serum glass vial where it was reconstituted with the assay media, MEM / EBBS (Minimum Essential Medium with Earle's Balanced Salt and supplemented with L-Glutamine and Non-Essential Amino Acid) to its target potency concentration by swirling until it was a homogeneous solution. An aliquot of the sample was then activated in 5 μg / mL trypsin diluted in assay media for one hour in a humidified incubator at 37° C., 5% CO2, then serially diluted four-fold in the assay media. The virus sample
example 2
Solutions for Preparation of Bioactive Stock Solutions
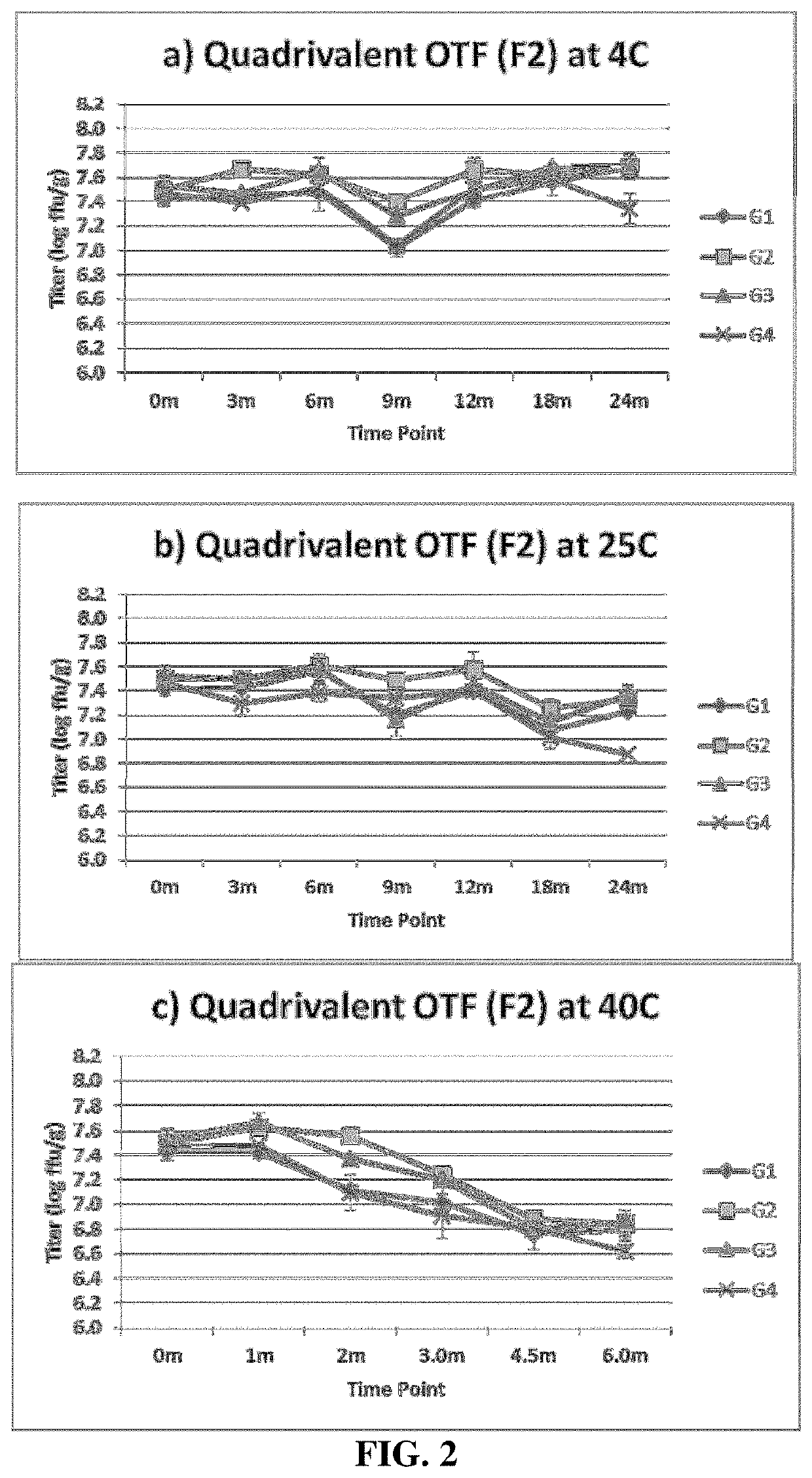
[0101]In the next several examples OTF's were fabricated using alternative processing conditions, compositions of film formers, and excipient profiles to investigate the impact on the process loss and storage stability of biologic potency of live rotavirus vaccines. A list of the chemical components used in the various excipient profiles tested is provided in Table 1.
TABLE 1Excipient profile of rotavirus stock solution formulationsprior to blending with polymer stock solution.ExcipientCitricPolySorbateProfileKPO4AcidSucroseSorbitolGlycerinCaCl2ZnCl2Gelatin80Designation(mM)(wt %)(wt %)(wt %)(wt %)(mM)(mM)(wt %)(wt %)F1500.8201.604440F2500.820004440F3500.820504440F4500.85004440F5500.820504400F6500.820500040F7500.85504440F8500.8201304440F9500.820064440F10500.820064400F11500.85064440F12500.820044440F13500.8200124440F14500.810064400F15500.85064400F16500.85504400F17500.851004400F185007.5000000F19500.820004420F20500.86004420F21500.82002544
example 3
hin Film Drying at Various Polymer Ratios
[0104]Following a similar approach to Example 2, live rotavirus-containing OTF's in the presence of different total concentration of pharmaceutical stabilizers, but with the same relative amounts, were evaluated for their ability to maintain potency through processing. The preparation methods are described below:
[0105]Live monovalent rotavirus vaccine was aseptically formulated at a titer of 6.5 log ffu / mL in the following pharmaceutical stabilizers: 4 mM zinc chloride, 4 mM calcium chloride, 0.8% solids content citric acid, 2% solids content gelatin, 50 mM potassium phosphate pH of 6.3, and 6% solids content sucrose (Formulation ‘F20’). In another container, the polymer mixture P10 was prepared as described in Example 2. Then, 1 to 8 parts of the formulated rotavirus solution was added to 19 parts of the polymer mixture to provide the values indicated in Table 3. This OTF wet blend was dispensed into a circular dish and dried for 3 hours in a
PUM
| Property | Measurement | Unit |
|---|---|---|
| Fraction | aaaaa | aaaaa |
| Fraction | aaaaa | aaaaa |
| Fraction | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap