Patents
Literature
Hiro is an intelligent assistant for R&D personnel, combined with Patent DNA, to facilitate innovative research.
13 results about "Organic compound" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
In chemistry, organic compounds are generally any chemical compounds that contain carbon. Due to carbon's ability to catenate (form chains with other carbon atoms), millions of organic compounds are known. The study of the properties, reactions, and syntheses of organic compounds comprises the discipline known as organic chemistry. For historical reasons, a few classes of carbon-containing compounds (e.g., carbonates and cyanide salts), along with a handful of other exceptions (e.g., carbon dioxide), are not classified as organic compounds and are considered inorganic. Other than those just named, little consensus exists among chemists on precisely which carbon-containing compounds are excluded, making any rigorous definition of an organic compound elusive.
CWB conductivity monitor
InactiveUS20120178175A1Low accuracyLow reliabilityComponent separationGeneral water supply conservationIon-exchange resinOrganic compound
This invention is a method and apparatus for monitoring the concentration of carbon dioxide dissolved in water by means of conductivity. It distinguishes between the conductivity resulting from carbon dioxide and the conductivity resulting from other constituents dissolved in water. It can be used to monitor the quality of demineralized water, boiler feedwater, steam, or condensate in electric power generation and other industrial facilities. It is constructed by adding a column containing weak base anion exchange resin and a conductivity instrument to a typical cation conductivity monitor. A sample of the water to be monitored flows first through a typical cation conductivity monitor, then through a weak base anion exchange column, and then through an additional conductivity instrument. Conductivity measured at the outlet of the weak base anion exchange column will be essentially due to whatever concentration of carbon dioxide is dissolved in the sample because other dissolved constituents that affect conductivity have been essentially removed by either the cation exchange resin that is part of a typical cation conductivity monitor, or by the weak base anion exchange resin. By subtracting the value of conductivity due to carbon dioxide (at the outlet of the weak base anion exchange column) from the value of cation conductivity (at the outlet of the cation exchange column), the value of degassed cation conductivity is obtained. In the title of the invention, CWB conductivity is an abbreviation for cation—weak base conductivity.In combination with existing methods for oxidizing organic compounds dissolved in water, this invention is also a method and apparatus for monitoring the concentration of dissolved or total organic carbon in water by means of conductivity. It distinguishes between the conductivity resulting from organic carbon and the conductivity resulting from inorganic constituents dissolved in water including carbon dioxide.
Owner:CROSMAN JAY CLIFFORD
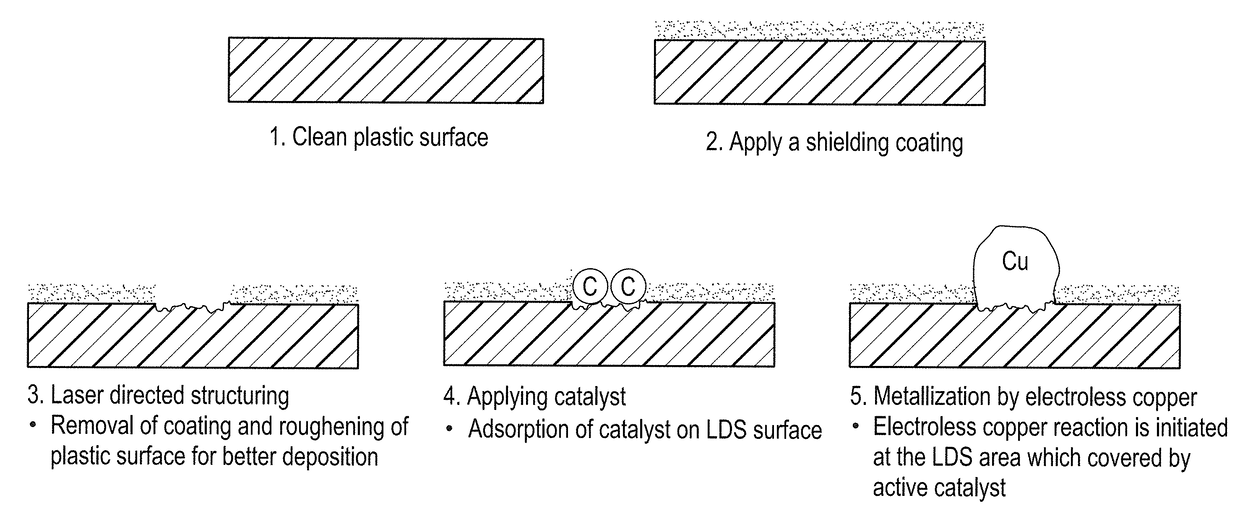
Shielding coating for selective metallization
InactiveUS9797043B1Avoid adsorptionLiquid/solution decomposition chemical coatingCable/conductor manufacturePolymer substrateOrganic compound
Owner:ROHM & HAAS ELECTRONICS MATERIALS LLC
Resin composition for laser engraving, relief printing plate precursor for laser engraving, relief printing plate and method of producing the same
The present invention provides a resin composition for laser engraving, including at least a complex between a layered inorganic compound and a cationic organic compound, and a binder polymer that is insoluble in water and soluble in an alcohol having 1 to 4 carbon atoms; a relief printing plate precursor for laser engraving using the same, a relief printing plate; and a method of producing the relief printing plate.
Owner:FUJIFILM CORP
Display device
ActiveUS20130134452A1High color purityImprove efficiencySolid-state devicesSemiconductor/solid-state device manufacturingCharge injectionPhysical chemistry
Owner:CANON KK
Reduction or elimination of microbial growth and biofouling of salted wet biomass byproducts
InactiveUS20070110909A1Negligibly corrosiveAvoid small quantitiesBiocideAntifouling/underwater paintsSimple Organic CompoundsSaline water
The invention describes a method, composition, and system of treating soluble organic compounds in combination with salts or brine used primarily for freezing point depression liquids, capable of aerobic and anaerobic microorganism growth such that by applying sufficient caustic in a caustic wash to an inside surface of any liquid media transport device for these freezing point depression liquids a caustic residue or caustic concentration associated with the caustic wash is created that results in mitigating or eliminating microorganism growth.
Owner:ISAKOFF LOUIS A +1
Conversion of biomass into a liquid hydrocarbon material
ActiveUS20190153324A1HydrogenOrganic-compounds/hydrides/coordination-complexes catalystsHydrogenGas phase
Owner:SHELL USA INC
Inhibitors of bacterial type iii secretion system
ActiveUS20140142134A1Effectively clearEfficient killingBiocideAntibacterial agentsEnteroinvasive E. coliBacteroides
Owner:MICROBIOTIX
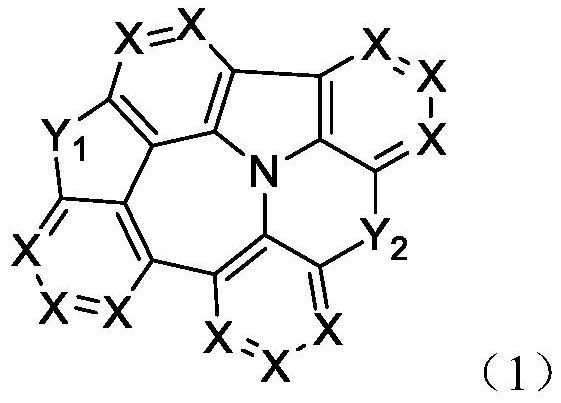
Organic compound containing nitrogen heterocyclic ring, mixture, composition and application
Owner:GUANGZHOU CHINARAY OPTOELECTRONICS MATERIALS
Efficient And Stable Inorganic Lead-Free Perovskite Solar Cell And Method For Preparing The Same
ActiveUS20220172904A1Good long-term work stabilityThe process is stable and efficientLight-sensitive devicesSolid-state devicesInorganic leadPerovskite solar cell
The disclosure provides an efficient and stable inorganic lead-free perovskite solar cell and a method for preparing the same. The solar cell includes a conductive substrate, a PEDOT: PSS layer, an inorganic lead-free CsSnI3 perovskite layer, a C60 layer, a BCP layer, and a metal counter electrode layer arranged in order from bottom to top, wherein the inorganic lead-free CsSnI3 perovskite layer is a CsSnI3 perovskite layer passivated by a thioureas small-molecule organic compound.
Owner:SHANDONG UNIV
Organic compound and organic light-emitting device using organic compound
PendingCN113292560AImprove thermal stabilityGood chemical propertiesOrganic chemistrySolid-state devicesArylSimple Organic Compounds
Owner:ZHEJIANG HUADISPLAY OPTOELECTRONICS CO LTD
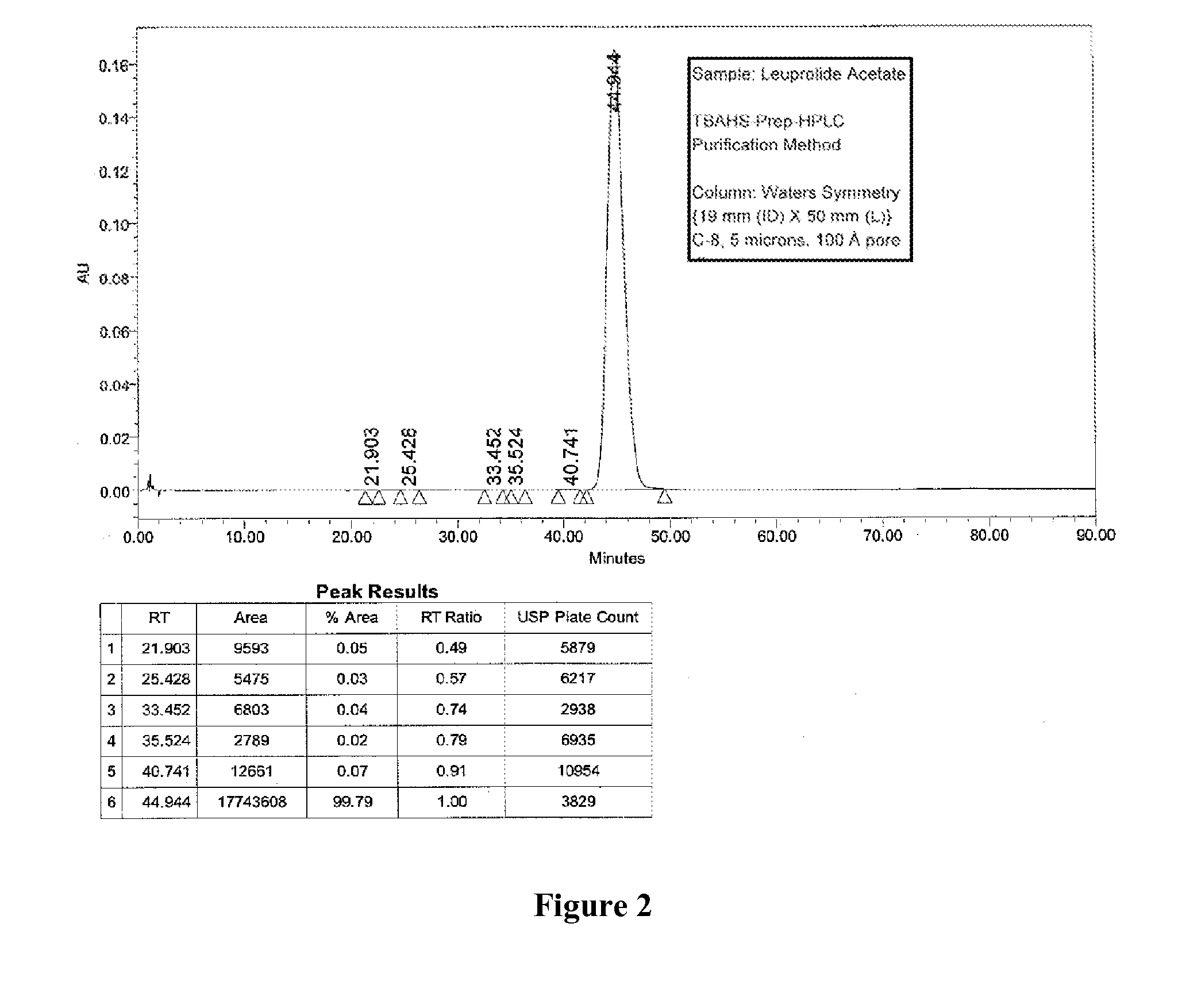
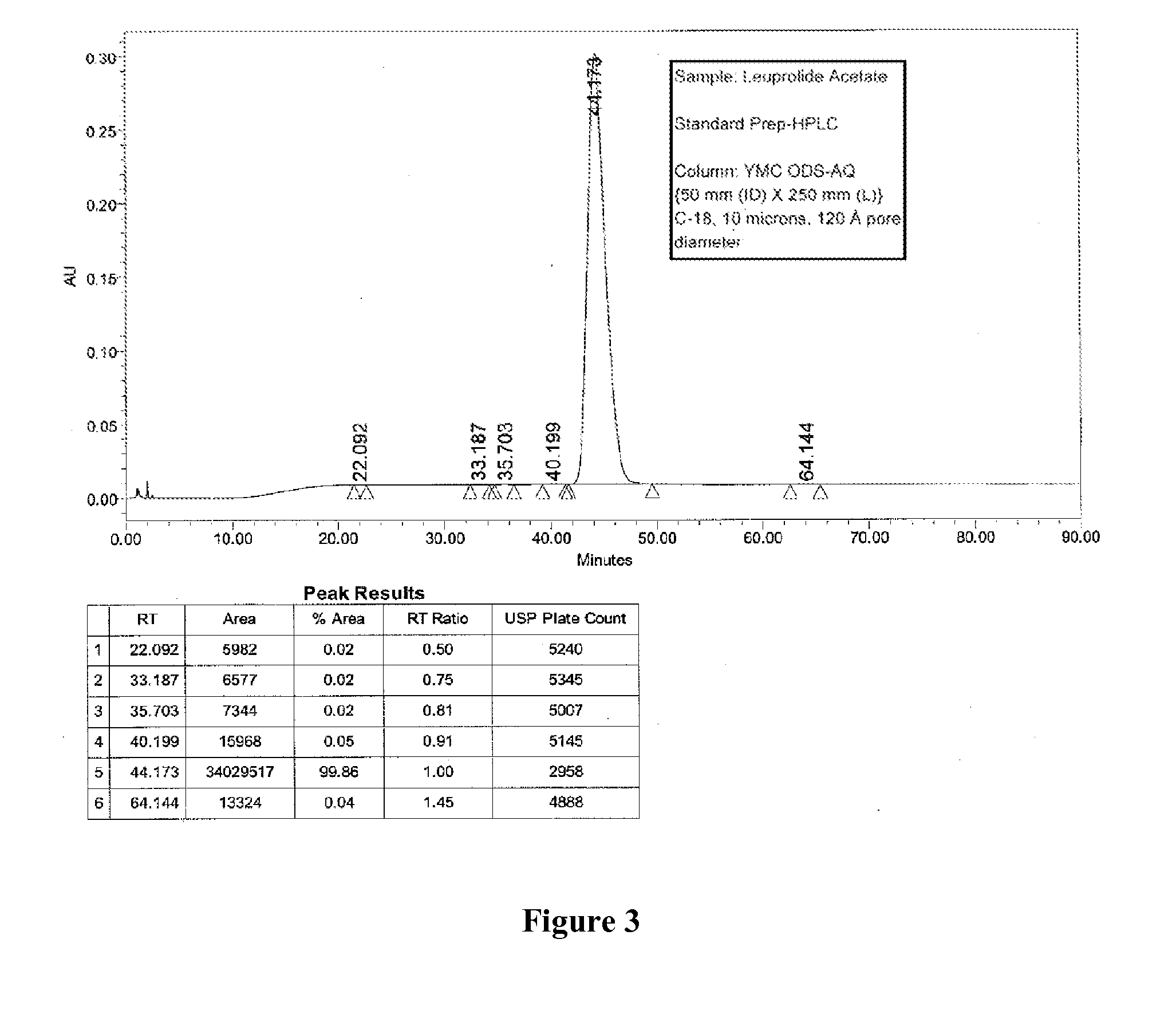
Purification of Organic Compounds Using Surrogate Stationary Phases on Reversed Phase Columns
ActiveUS20150360146A1Easy loadingLimited use of solventCation exchanger materialsComponent separationStationary phasePhosphonium salt
Owner:NEULAND HEALTH SCI
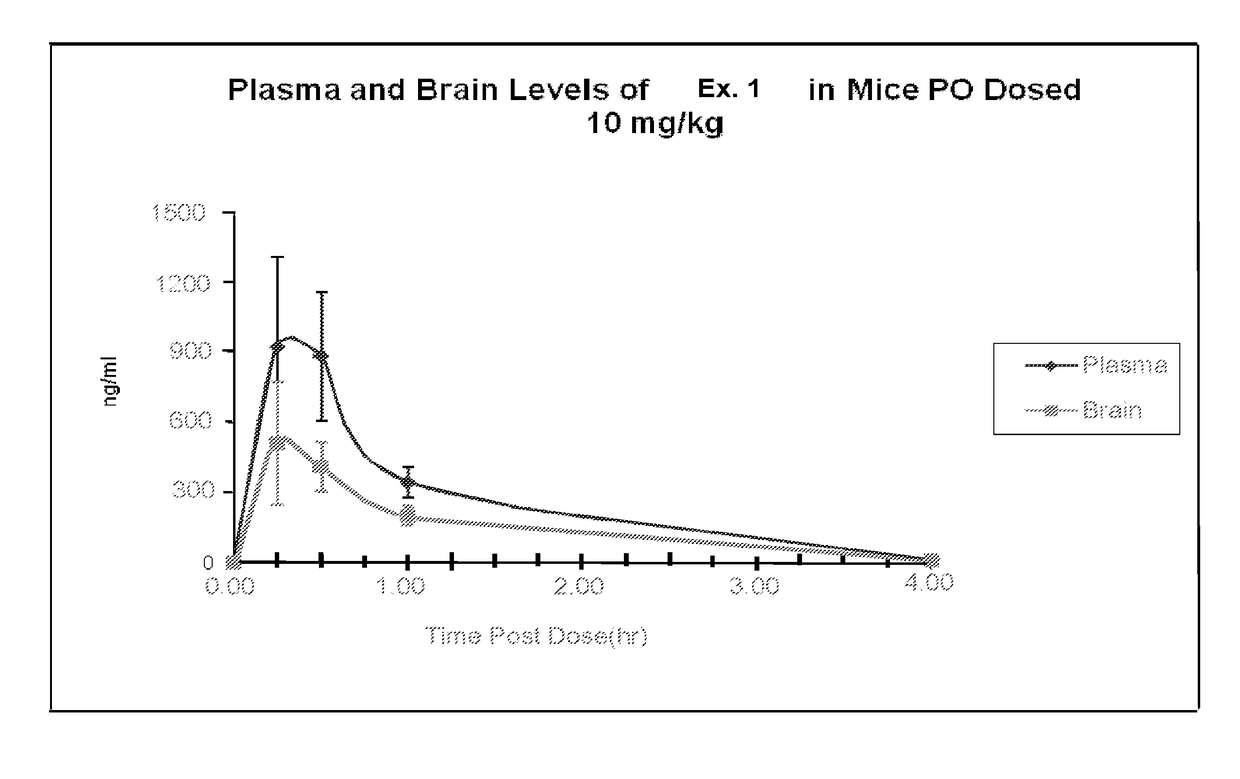
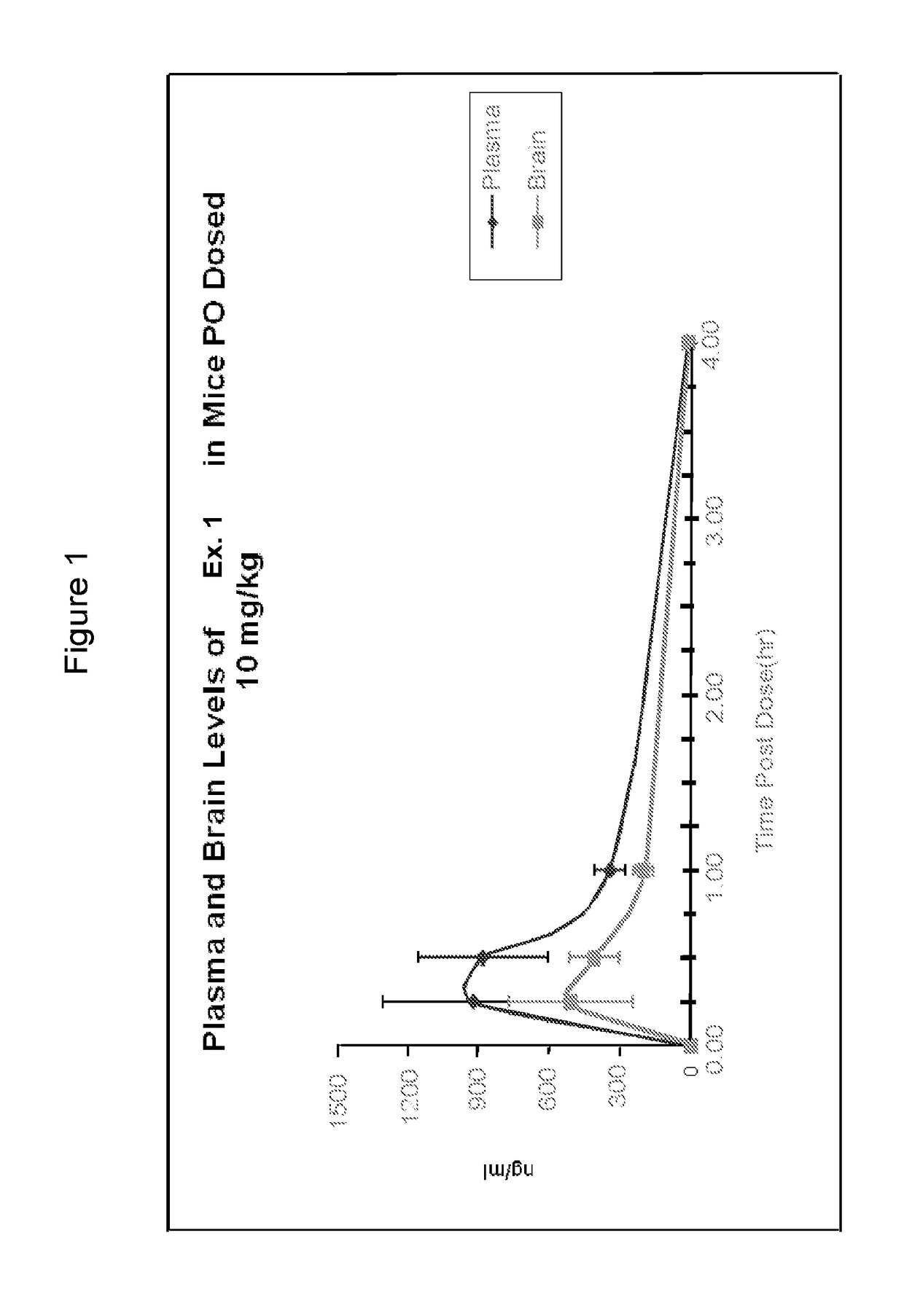
Organic compounds
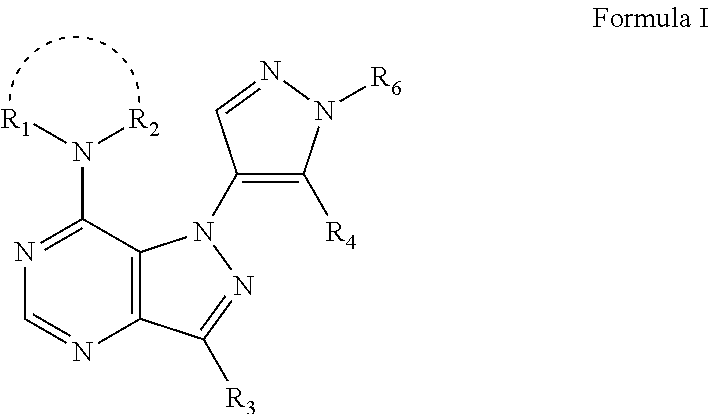
ActiveUS20170360792A1Potent and selective PDE inhibitory propertyImproved orally availabilityOrganic active ingredientsNervous disorderOrganic compoundMedicinal chemistry
Owner:INTRA CELLULAR THERAPIES INC
Who we serve
- R&D Engineer
- R&D Manager
- IP Professional
Why Eureka
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Social media
Try Eureka
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap