Patents
Literature
Hiro is an intelligent assistant for R&D personnel, combined with Patent DNA, to facilitate innovative research.
40results about How to "Improve solubility" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
Copolymer, process for producing the same, and resist composition
InactiveUS6706826B1Improve adhesionImprove solubilityOriginals for photomechanical treatmentPhotosensitive materials for photomechanical apparatusSolventResist
The present invention relates to a copolymer for use in paints, resists, and the like; a method for manufacturing the same; and a resist composition using the same. The copolymer according to the present invention is obtained by means of polymerizing at least one monomer containing an alicyclic structure and one monomer containing a lactone structure, and the distribution of the copolymer composition of said monomer containing a lactone structure in said copolymer is in the range of -10 to +10 mol % of the average copolymer composition of said monomer containing a lactone structure in said entire copolymer. In addition, the copolymer according to the present invention is obtained by means of polymerizing a monomer containing an alicyclic structure, a monomer containing a lactone structure, and another vinyl monomer comprising a higher polarity than said monomer containing an alicyclic structure, but a lower polarity than said monomer containing a lactone structure. The copolymer according to the present invention exhibits superior adhesion properties to surfaces possessing a high polarity, such as metal surfaces and the like, in addition to excellent hydrophobic and thermal resistance properties, and also displays a favorable solubility in solvents used for paints, resists, and the like.
Owner:MITSUBISHI CHEM CORP
Uniform delivery of topiramate over prolonged period of time with enhanced dispersion formulation
InactiveUS20050058707A1Improve bioavailabilityPromote absorptionNervous disorderCapsule deliveryTopiramateControlled Release Dosage Form
Compositions and dosage forms for enhanced dispersion of topiramate in a controlled release dosage form delivered as a dry or substantially dry erodible solid at a uniform rate over a prolonged period of time are described.
Owner:ALZA CORP
Crystal form G of ibrutinib and preparation method
InactiveCN105646499AImprove stabilityHigh purityOrganic active ingredientsOrganic chemistry methodsSolubilitySolvent
The invention discloses a crystal form G of ibrutinib. The crystal form G is characterized in that X-ray powder diffraction (X-RPD) which adopts Cu-Kalpha radiation and is represented with a 2theta angle has diffraction peaks in positions at angles of 5.0 degrees plus or minus 0.2 degrees, 7.3 degrees plus or minus 0.2 degrees, 10.1 degrees plus or minus 0.2 degrees, 12.0 degrees plus or minus 0.2 degrees, 13.2 degrees plus or minus 0.2 degrees, 17.1 degrees plus or minus 0.2 degrees, 19.5 degrees plus or minus 0.2 degrees, 20.8 degrees plus or minus 0.2 degrees, 22.3 degrees plus or minus 0.2 degrees, 24.3 degrees plus or minus 0.2 degrees, 27.4 degrees plus or minus 0.2 degrees and 31.2 degrees plus or minus 0.2 degrees. Related solvents in a preparation process of the crystal form G are cheap, the conditions are mild, the operation is simple, good controllability and reproducibility are realized, further, the prepared crystal form has great stability, the HPLC (high performance liquid chromatography) purity is higher than 99%, and the phenomenon of crystal transformation can be avoided; besides, the solubility is high, the dissolubility is good, and the bioavailability is high.
Owner:孙霖
Analogs and prodrugs of buprenorphine
InactiveUS20040192714A1Improve lipophilicityImprove solubilityBiocideOrganic chemistryBuprenophineProdrug
Owner:PURDUE PHARMA LP
Method of recovering and regenerating waste gas desulfurizing and defluorinating absorbent liquid continuously
ActiveCN1974430ALow investment costLow running costDispersed particle separationWater/sewage treatmentSulfite saltSodium bisulfite
The method of recovering and regenerating waste gas desulfurizing and defluorinating absorbent liquid continuously includes absorbing SO2 and / or fluoride from waste gas with alkaline absorbent to generate desulfurizing and defluorinating absorbent liquid containing sodium sulfite, sodium bisulfite and / or fluoride; the subsequent reacting the desulfurizing and defluorinating absorbent liquid with magnesium-base suspension or solid to obtain magnesium sulfite and / or magnesium fluoride precipitate; filtering to eliminate magnesium sulfite and / or magnesium fluoride precipitate, and recovering the filtrate at pH 6.5-9.5 for reuse. The said method has fast regeneration reaction speed, low cost and complete reaction.
Owner:陆泳凯 +1
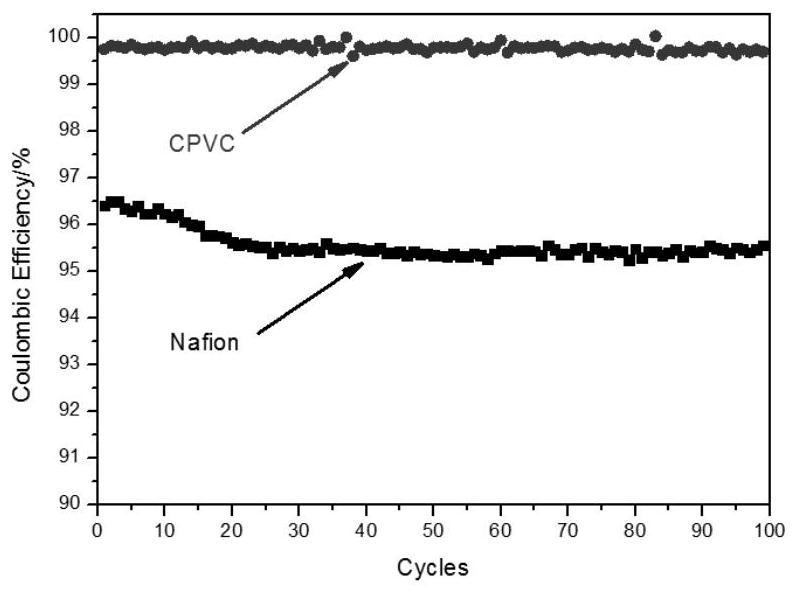
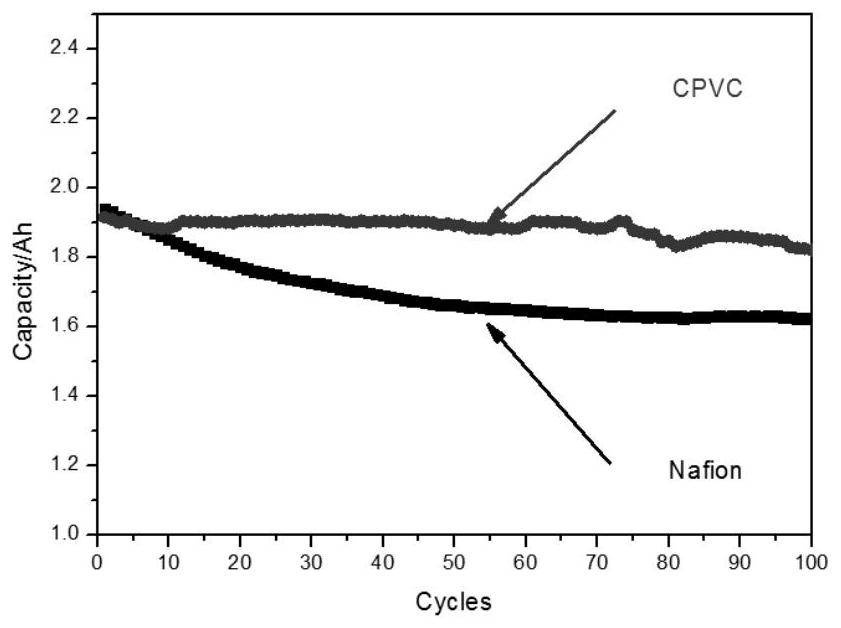
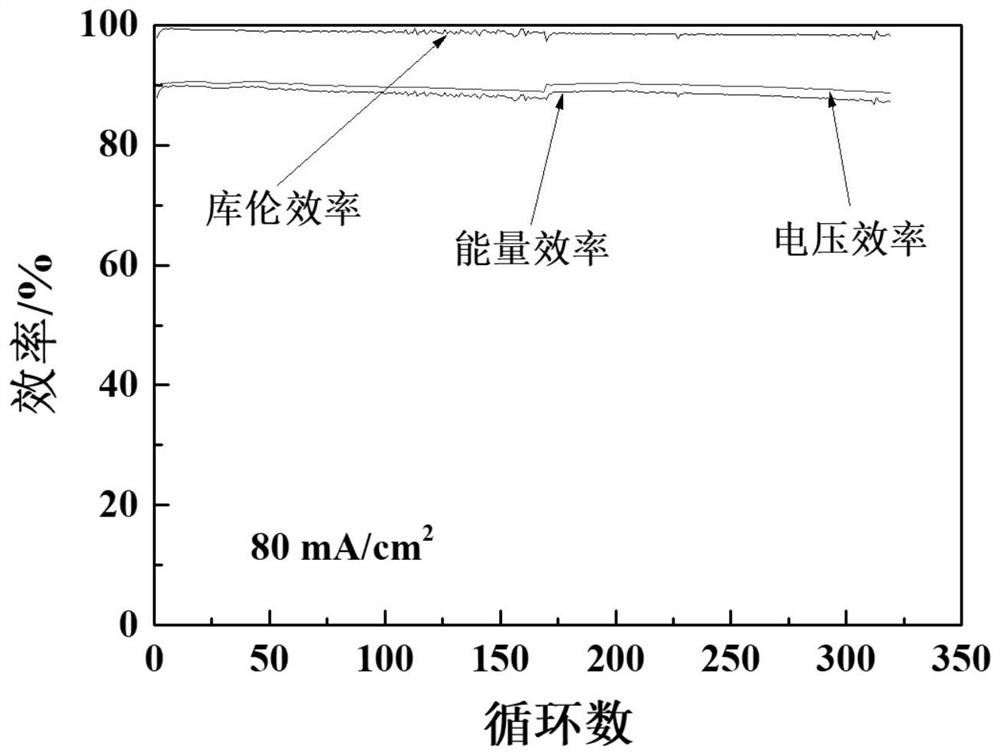
Application of ion conduction membrane containing chlorinated polyvinyl chloride in flow battery
ActiveCN112447994AHigh chemical stabilityImprove solubilityRegenerative fuel cellsAqueous electrolyte fuel cellsIon exchangeSolvent evaporation
Owner:DALIAN INST OF CHEM PHYSICS CHINESE ACAD OF SCI
Titanium-series polyester catalyst and method thereof for producing copolyester
Owner:JIANGSU JINGHONG NEW MATERIAL TECH
Method for improving degree of exhaustion during wood dyeing
InactiveCN105643745APressure wood treatmentWood treatment detailsAcetone / EthanolSupercritical fluid extraction
The invention discloses a method for improving the degree of exhaustion during wood dyeing and belongs to the technical field of wood dyeing. The method specifically comprises the following steps: adjusting the moisture content of wood to be 50-100%, carrying out supercritical CO2 fluid extraction by adopting a mixed solution containing acetone, ethanol and one or more fatty alkyl tertiary amines as an entrainer, and then carrying out pressure relief at a rate of 1.0-10.0 MPa / min to obtain pretreated wood; soaking the pretreated wood in a mixed solution containing one or more active dye liquids and sodium sulfate to obtain dyed wood; and conducting fixation and soaping on the dyed wood. The method is simple in process, and has the advantages that not only can the degrees of exhaustion and fixation of an active dye in wood be improved, but also the dyeing depth can be improved.
Owner:CENTRAL SOUTH UNIVERSITY OF FORESTRY AND TECHNOLOGY
Special-purpose herbal antipersprant deodorant for sensitive skin and preparation method thereof
InactiveCN104523469ASuitable for useNo artificial colors addedCosmetic preparationsToilet preparationsHamamelis virginianaPolyphenol
The invention provides a special-purpose herbal antipersprant deodorant for sensitive skin and a preparation method thereof. The herbal antipersprant deodorant for sensitive skin comprises the following components by weight percentage: 0.03-3% of honeysuckle flower extract product, 0.02-2% of wrinkled giant hyssop extract product, 0.5-3% of wild chrysanthemum flower extract product, 0.5-3% of witch hazel extract product, 0.7-4% of lavender extract product, 0.05-5% of tea polyphenol, 5-10% of propylene glycol, 0.03-3% of essence, 0.02-3% of mint, 0.5-5% of carragheenan and the balance of deionized water. The herbal antipersprant deodorant has the efficacy for inhibiting secretion of sweat and bacteria growth, generation of body odour can be avoided, clothes wetted by sweat can be avoided, Chinese herbal medicine components are safe and natural, so that skin can not be stimulated, skin problem due to sweat can be alleviated, and the special-purpose herbal antipersprant deodorant can be used for population with sensitive skin.
Owner:AESTHETIC TECH BEIJING
Probiotics plant solid beverage for treating hyperuricemia and gout
InactiveCN110623182APreserve nutritional valueDisease resistantLactobacillusSkeletal disorderFreeze dryProbiotic
The invention provides a probiotics plant solid beverage for treating hyperuricemia and gout. The solid beverage is prepared by mixing freeze-dried probiotics powder and plant extract with the addition of proper auxiliary materials. The solid beverage comprises the following components: the plant extract, the auxiliary materials and the probiotics, with the sum of the mass percentage of each component being 100%. The plant extract comprises celery seed powder, sophora flower bud powder and platycodon root powder. The invention has an obvious effect on lowering uric acid and relieving gout.
Owner:胡洁
Anti-aging folia perillae acutae oil nano-emulsion cosmetic
InactiveCN102166175AImprove solubilityImproves free radical scavengingCosmetic preparationsToilet preparationsSolubilitySkin permeability
The invention discloses an anti-aging folia perillae acutae oil nano-emulsion cosmetic. The grain diameter of the cosmetic is 6-100nm. The anti-aging cosmetic comprises the following raw materials: 15.00-31.00wt% of surfactant, 4.00-7.00wt% of oil, 0.10-10.00wt% of folia perillae acutae oil, 1.00-2.00wt% of vitamin E acetic ester, 0.10-0.20wt% of essence and the balance of distilled water, wherein the total amount is equal to 100%. The cosmetic has better skin permeability, can enhance the solubility of the folia perillae acutae oil, improves the anti-aging effect of the folia perillae acutae oil, improves the ductility of the cosmetic on the skin, and brings clear and moisture good feeling for users. The anti-aging cosmetic has wide market prospect in the field of the anti-aging cosmetics.
Owner:杨平
Method for preparing alumina-based porous mineral material
ActiveCN102188950AImprove adsorption capacityLarge specific surface areaOther chemical processesWater/sewage treatment by sorptionKaoliniteWastewater
The invention discloses a method for preparing an alumina-based porous mineral material, which comprises the following steps of: grinding kaolinite with the purity of not less than 90 percent into 70 to 80 mass percent of fine particles with the particle size of -100 meshes; calcining the kaolinite fine particles at the high temperature of between 1,050 and 1,150 DEG C for 10 to 30 minutes; extracting silica from the calcined kaolinite by using 100 to 140g / L sodium hydroxide solution in a solid-liquid mass ratio of 1:8-1:12 at the temperature of between 90 and 140 DEG C for 90 to 120 minutes with stirring; and performing solid-liquid separation, and drying the obtained solid to obtain the alumina-based porous mineral material which has a good effect of adsorbing heavy metal ions in solution. The alumina-based porous mineral material prepared by the method is prepared from natural minerals with wide sources and large resource reserves, can be used for treating heavy metal ion polluted wastewater, and has a wide application prospect in the environment-friendly industry, and the preparation process is simple, and low in cost.
Owner:CENT SOUTH UNIV
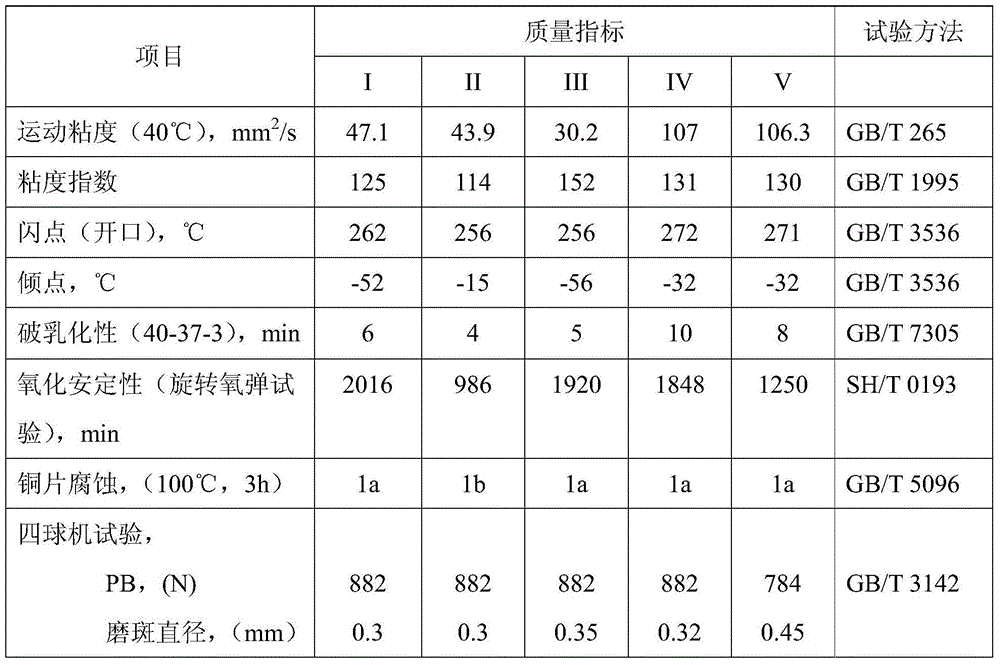
Air compressor oil composition and preparing method thereof
ActiveCN103820192AImprove lubricity and wear resistanceImprove solubilityLubricant compositionEmulsionAir compressor
Owner:SHANGHAI HITECRUN SPECIAL LUBRICATE SCI & TECH
Water-soluble hypocrellin PLGA nanoparticle and preparation method thereof
InactiveCN103933568AGood biocompatibilitySmall particlesPowder deliveryEnergy modified materialsSolubilitySide effect
The invention relates to the technical field of medicines, and particularly relates to a water-soluble hypocrellin PLGAnanoparticle and a preparation method thereof. The preparation method comprises the following steps: by taking poly(lactic-co-glycolic acid) (PLGA) as a carrier, dissolving PLGA and hypocrellin into an organic solvent to prepare an oil phase, adding into freeze-drying excipient-containing water phase under high-speed stirring, and finally obtaining the hypocrellin nanoparticle by adopting an emulsification freeze-drying method. By using a biodegradable medical polymer material PLGA used in the preparation method and approved by the food and drug administration (FDA) of the United States, the toxic and side effects of a common drug-carrying material can be reduced, the water solubility of the drug can be improved, the particle size of the prepared nanoparticle ranges from 20-200nm, the drug has red shift, the absorption of the nanoparticle in a phototherapy window (600-900nm) can be increased, and the dark toxicity of the drug can be reduced. The preparation method is simple in raw materials and easy to operate.
Owner:NANJING NORMAL UNIVERSITY
Hyaluronic acid-based modified gliadin nanoparticles as well as preparation method and application thereof
ActiveCN112120212AImprove solubilityGood emulsifying effectPowder deliveryCosmetic preparationsGlycerolPickering emulsion
The invention discloses hyaluronic acid-based modified gliadin nanoparticles as well as a preparation method and application thereof. The method for preparing the W2 / O2 / (O1 / W1) type multiple emulsionfrom the nanoparticles comprises the following steps: mixing polyglycerol ricinoleate with corn oil at 65 DEG C to form a first oil phase, mixing a mixed solution of sodium chloride and gelatin with the obtained first oil phase, and performing homogenizing by adopting high-pressure microjet to obtain a W2 / O2 type primary emulsion; mixing a second oil phase with the nanoparticle suspension, and performing high-shear treatment to obtain an O1 / W1 type Pickering emulsion; and mixing the W2 / O2 type primary emulsion with the O1 / W1 type Pickering emulsion by adopting a high-pressure homogenization technology, so as to prepare the W2 / O2 / (O1 / W1) type multiple emulsion. The preparation method of the multiple emulsion is simple, mild in reaction condition and suitable for industrial production, and can be applied to the fields of foods, medicines, cosmetics and the like.
Owner:HEFEI UNIV OF TECH
Separation method of waste polyphenylene sulfide and polytetrafluoroethylene blended dust settling pockets
The invention relates to a separation method of waste polyphenylene sulfide and polytetrafluoroethylene blended dust settling pockets. The separation method comprises the steps that the waste polyphenylene sulfide and polytetrafluoroethylene blended dust settling pockets are firstly smashed, then the smashed mixed materials are added into a separating medium with the density between the density of polyphenylene sulfide and the density of polytetrafluoroethylene for centrifugal separation, blended fabric is smashed according to the different densities of two fibers, and the aim of recycling polyphenylene sulfide and polytetrafluoroethylene in the waste dust settling pockets is achieved through centrifugal separation. The purity of polyphenylene sulfide materials obtained after separation reaches 97% or above, the purity of polytetrafluoroethylene materials reaches 91% or above, the material separation purity is high, the separation effect is good, and the recycling value is high.
Owner:ANHUI YUANCHEN ENVIRONMENTAL PROTECTION SCI & TECH
Xinjiang-style grease, and preparation method of micro-emulsion thereof
InactiveCN105853258AChange oil solubilityReduce the degree of oxidationCosmetic preparationsToilet preparationsSolubilityAntioxidant
The invention discloses a Xinjiang-style grease micro-emulsion and a preparation method thereof. The preparation method comprises the following steps: 1, shredding Xinjiang-style fruits; 2, squeezing the shredded fruits, and collecting squeezed crude oil and oil cakes; 3, filtering the crude oil obtained in step 2 to obtain clarified Xinjiang-style grease; and 4, uniformly stirring the Xinjiang-style grease, lecithin, tween65, ethylene glycol distearate, sodium N-lauroylsarcosinate, dodecyl glycoside, lauroyl diethanolamine, sodium dioctylsulfosuccinate, isooctyl palmitate, an antioxidant, glycerin and water to prepare the Xinjiang-style grease micro-emulsion. The above system changes the oil solubility of Xinjiang-style grease, increases the solubility, and reduces the oxidation degree and instability of the Xinjiang-style grease; and compared with traditional emulsions, the stable micro-emulsion prepared in the invention has the advantages of small particle size, uniform dispersion, high stability and convenient storage.
Owner:INST OF AGRI MECHANIZATION XINJIANG AGRI INST
Desulfurization synergist for calcium-based wet desulfurization, preparation and application method thereof
InactiveCN108176217AImprove the safety of useAvoid direct dischargeGas treatmentDispersed particle separationCarboxyl radicalPhysical chemistry
Relating to the technical field of desulfurization synergists, the invention discloses a desulfurization synergist for calcium-based wet desulfurization, and a preparation and application method thereof. The desulfurization synergist comprises 50-70% of an acrylic copolymer and 30-50% of a polysaccharide compound by mass percentage. The acrylic copolymer is prepared by copolymerization reaction ofa carboxyl-containing monomer and an inert monomer in a mass ratio of 1:2-5. The desulfurization synergist provided by the invention has simple composition, can effectively improve the desulfurization efficiency, reduce the calcium-sulfur ratio of calcium-based wet desulfurization, lower the operation energy consumption of the desulfurization system, and ensure the system stability and the purityof the gypsum by-product, and effectively avoids some small molecule substances in the composition from entering the atmosphere along with flue gas.
Owner:BEIJING GUODIAN LONGYUAN ENVIRONMENTAL ENG
Manufacturing technology of curtain fabric capable of releasing negative ions
InactiveCN111893755AConducive to fixationUniform fixationCurtain accessoriesWoven fabricsFlax fiberChemistry
The invention discloses a manufacturing technology of a curtain fabric capable of releasing negative ions. The manufacturing technology comprises the following steps that S1, flax fibers and polyesterfibers are blended into warps and wefts; S2, the warps and the wefts are woven into a main fabric through warp-weft knitting; S3, bleaching is carried out, wherein the main fabric obtained in S2 is bleached; S4, a finishing liquid is prepared, and the main fabric bleached in S3 is added into the finishing liquid and subjected to dipping; S5, residual liquid is discharged, wherein the residual liquid of the main fabric subjected to dipping in S4 is discharged; and S6, the main fabric without the residual liquid is dried to obtain the main fabric capable of releasing the negative ions. The finishing liquid in S4 comprises, by weight, 6-7 parts of tourmaline ultra-fine powder, 5-7 parts of seaweed carbon ultra-fine powder; 3-4 parts of dispersant, 2-4 parts of a fixing agent, 1-2 parts of apermeating agent and 30-40 parts of deionized water. The curtain fabric has the function of releasing the negative ions, so that volatile gases such as formaldehyde and ammonia in new furniture can beeliminated, and the damage of gases such as formaldehyde to the human body can be reduced.
Owner:SHANGHAI YUQI IND CO LTD
Preparation method of poultry plasma protein
ActiveCN108813090ACompact structureChange in structural complianceProtein composition from bloodProtein foodstuffs working-upMedicinePlasma lipids
The invention discloses a preparation method of poultry plasma protein. The preparation method comprises the following steps of step I, taking poultry blood, and performing treatment to obtain plasmalipid; step II, performing ultrasonic treatment on the plasma lipid, wherein the power for ultrasonic treatment is 100-300W, and the time for ultrasonic treatment is 10-30min; step III, regulating thepH of the plasma lipid after ultrasonic treatment to 8.5-9.5, then performing ultrafiltration under the ultrafiltration pressure of 0.3-0.6MPa, cutting off the plasma lipid of which the molecule is 5000-10000Da, wherein the ultrafiltration circulation time is 10-30min; and step IV, drying the plasma lipid obtained in the step III, so as to obtain the poultry plasma protein powder. The solubilityof the poultry plasma protein prepared by the method disclosed by the invention is increased to 85-88% from 50-56%. The plasma protein prepared by the method has better antioxidant activity.
Owner:JIANGSU ACAD OF AGRI SCI
Flavoring agent
InactiveCN102239961AImprove solubilityAvoid cloggingAnimal feeding stuffAccessory food factorsNutrientMaltodextrin
Owner:重庆味来香生物科技有限公司
Fluorine-containing highly branched polymer and epoxy resin composition containing the same
ActiveUS20150158959A1Increased modificationLess tanglePlastic/resin/waxes insulatorsEpoxy resin coatingsPolymer scienceOxetane
A fluorine-containing highly branched polymer obtained by polymerizing a polyfunctional monomer A that has two or more radically polymerizable double bonds and all or a portion of which has a bisphenol structure, a monomer B having a fluoroalkyl group and at least one radically polymerizable double bond, within a molecule, and a monomer C having at least one ring-opening polymerizable group selected from the group including an epoxy group and an oxetanyl group, and having at least one radically polymerizable double bond, within a molecule, under the presence of a polymerization initiator D with an amount of 5% by mole to 200% by mole to the number of moles of the polyfunctional monomer A; an epoxy resin composition including the polymer; and an epoxy resin cured product obtained from the resin composition.
Owner:NISSAN CHEM CORP
Molding and foaming process and device of macromolecule with supercritical rapid swelling through mold pressing
PendingCN109955424AFacilitated physical diffusion rateImprove solubilityInjection molding machineMelt blending
Owner:JIANGSU JICUI ADVANCED POLYMER MATERIAL RES INST CO LTD +1
Environment-friendly water-based ink cleaning agent and preparation method thereof
Aiming at the disadvantages that the cleaning effect of water-based ink cleaning agents on the current market is not ideal, the surface of cleaned workpiece is corroded by added strong alkaline assistants, the environment is easily polluted and the like, the invention provides an environment-friendly water-based ink cleaning agent which has high cleaning efficiency, does not easily corrode the surface and is easily biodegrade, and a preparation method of the ink cleaning agent. The water-based ink cleaning agent comprises the following components in percentage by mass: 5-10% of an anionic-cationic surfactant compound, 6%-15% of a polar solvent, 2%-5% of a solubilizer, 5%-10% of other auxiliary agents and the balance of water.
Owner:NANJING UNIV OF INFORMATION SCI & TECH
Preparation method of traditional Chinese medicine granules for treating pig wind-heat type common cold
InactiveCN111588750AGreat tasteImprove solubilityPharmaceutical non-active ingredientsGranular deliveryBiotechnologyCommon cold
The invention discloses a preparation method of traditional Chinese medicine granules for treating pig wind-heat type common cold. The preparation method comprises the following steps of 1) performingmixing on a traditional Chinese medicine extract and auxiliary materials according to a ratio of 1: (4.5-6.5), performing grinding and crushing until the mixture is uniformly mixed to obtain a mixture, wherein the traditional Chinese medicine extract comprises the following components: 60-68g of honeysuckle extract; 27 to 35 g of scutellaria baicalensis extract; 40 to 46 g of licorice extract; 2)adding a wetting agent into the mixture obtained in the step 1), performing mixing, and uniformly stirring to obtain formed particles, wherein the use amount of the wetting agent is 15-25% of the mass of the mixture; and 3) performing drying on the formed particles obtained in the step 2) at the temperature of 50-60 DEG C to obtain the traditional Chinese medicine particles. According to the scheme, proper auxiliary materials are added for taste correction, and the traditional Chinese medicine particles with better water solubility are prepared through the wetting agent, so that clinical application is facilitated.
Owner:GUANGDONG RONGDA BIOENG CO LTD
Leimaquban solid dispersion, prepation method and oral composition
ActiveCN1533768AImprove bioavailabilityImprove solubilityCapsule deliveryDrageesActive agentActive component
Owner:ADVENCHEN LAB NANJING
Whole-vegetable-based high-energy ready-to-drink nourishment and preparation method thereof
The invention discloses a whole-vegetable-based high-energy ready-to-drink nourishment and a preparation method thereof. The preparation method comprises the following steps: firstly, adding water into vegetable protein to prepare vegetable protein dispersion liquid, carrying out hydrodynamic cavitation treatment on the vegetable protein dispersion liquid in a cavitation cavitation machine, then adding vegetable fat, carbohydrate, vitamins, mineral substances and residual water, homogenizing to form emulsion, introducing the emulsion into two scraper heat exchangers which are connected in series, shearing and granulating in the first scraper heat exchanger, and shearing and cooling in the second scraper heat exchanger to obtain the full-vegetable-based high-energy ready-to-drink nourishment. The obtained nutriment is a full-vegetable-based high-energy-density nutriment which is relatively high in protein and fat content and relatively comprehensive in nutritional ingredients, is green, safe, nutritional and healthy, has the characteristics of low viscosity and good storage stability, and can meet basic energy requirements of patients with eating obstacles.
Owner:SOUTH CHINA UNIV OF TECH
Preparation method of cleaning agent for cleaning and drying loading arm steering joint
InactiveCN105462716AImprove processing efficiencyReasonable formula designOrganic non-surface-active detergent compositionsEmulsionEngineering
The invention relates to the technical field of loading arm cleaning and maintenance and particularly relates to a preparation method of a cleaning agent for cleaning and drying a loading arm steering joint. The method comprises the following steps: adding 15 parts of acetone, 5 parts of acrylate emulsion and 25 parts of water into a container, and stirring for 5 minutes; slowly adding 5 parts of citric acid and stirring at the same time, and after the addition, stirring for 5 minutes; slowly adding 5 parts of ethyl ether and 15 parts of water and stirring at the same time, and after the addition, stirring for 5 minutes; slowly adding 0.5 part of rhamnoside and stirring at the same time, and after the addition, stirring for 10 minutes; and finally, quickly adding 10 parts of cyclohexane and 15 parts of water and stirring for 10 minutes. According to the preparation method of the cleaning agent provided by the invention, through gradual addition and different adding sequences, the dissolving property among the substances can be improved, and the possibility of particle aggregation can be avoided so as to remarkably improve the treatment efficiency of the cleaning agent.
Owner:WUHU JINYU PETROCHEM EQUIP
Treatment method of fermentation antibiotic wastewater
ActiveCN109052720AImprove solubilityReduce concentrationTreatment using aerobic processesWater contaminantsSludgeAntibiotic Y
The invention belongs to the technical field of water, wastewater, sewage or sewage treatment, and in particular discloses a treatment method of fermentation antibiotic wastewater. The method comprises a pretreatment stage and a biochemical treatment stage, wherein the pretreatment stage comprises the following steps: (1) removing suspended solids; (2) adding quicklime into the wastewater treatedin step (1) so as to enable the pH value of the wastewater to be equal to 8-10; (3) adding a cationic polymer into the wastewater treated in the step (2); (4) filtering the wastewater, treated in thestep (3), with a filter membrane; (5) adding concentrated sulfuric acid into the wastewater treated in the step (4) until the pH value of the wastewater is equal to 2-3; (6) adding the quicklime intothe wastewater treated in the step (5) so as to enable the pH value of the wastewater to be equal to 6-8; (7) settling and filtering the wastewater treated in the step (6). The method not only can enable antibiotics to inactivate and does not cause secondary pollution, but also can cause partial ions, generated after hydrolysis of the antibiotics, to precipitate so as to effectively reduce the concentration of the wastewater.
Owner:CHONGQING TECH & BUSINESS UNIV
Quality detection method of patchouli oil, clathrate compound of patchouli oil, dry suspension and application of clathrate compound and dry suspension
PendingCN113533603AQuick checkEasy to storeComponent separationDigestive systemGuaienePharmacometrics
The invention relates to a quality detection method of patchouli oil, clathrate compound of patchouli oil, dry suspension and pharmaceutical application of the clathrate compound anddry suspension. The quality detection method of the patchouli oil comprises the following steps o: S1, measuring the spectrum of the patchouli oil by using GC-MS; S2, matching the spectrum in the step S1 according to a GC-MS NIST database to obtain qualitative affiliation information of characteristic peaks; and S3, according to the spectrum in the step S1 and the qualitative affiliation information in the step S2, detecting the peak areas corresponding to actin, delta-guaiacol, patchouli alcohol and patchouli ketone in the spectrum in the step S1, and if the ratio of the sum of the peak areas is greater than 70%, indicating that the patchouli oil is qualified. According to the quality detection method of the patchouli oil, the quality of traditional Chinese medicine can be rapidly detected, traditional Chinese medicine standards are standardized, pharmacological research of traditional Chinese medicine is promoted, and a safe, reliable and convenient-to-store traditional Chinese medicine preparation is provided.
Owner:SHANGHAI INST OF MATERIA MEDICA CHINESE ACAD OF SCI
Who we serve
- R&D Engineer
- R&D Manager
- IP Professional
Why Eureka
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Social media
Try Eureka
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap